Input interpretation

titanium tetrachloride
Chemical names and formulas

formula | TiCl_4 Hill formula | Cl_4Ti name | titanium tetrachloride IUPAC name | tetrachlorotitanium alternate names | tetrachlorotitanium | titanic chloride | titanium chloride | titanium(IV) chloride mass fractions | Cl (chlorine) 74.8% | Ti (titanium) 25.2%
Structure diagram
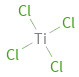
Structure diagram
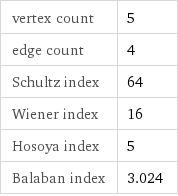
vertex count | 5 edge count | 4 Schultz index | 64 Wiener index | 16 Hosoya index | 5 Balaban index | 3.024
Basic properties
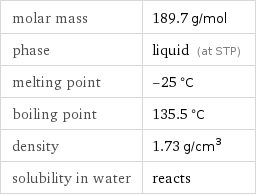
molar mass | 189.7 g/mol phase | liquid (at STP) melting point | -25 °C boiling point | 135.5 °C density | 1.73 g/cm^3 solubility in water | reacts
Units

Liquid properties (at STP)
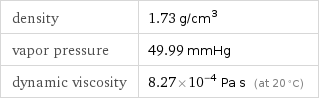
density | 1.73 g/cm^3 vapor pressure | 49.99 mmHg dynamic viscosity | 8.27×10^-4 Pa s (at 20 °C)
Units
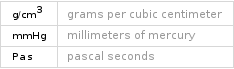
Thermodynamic properties
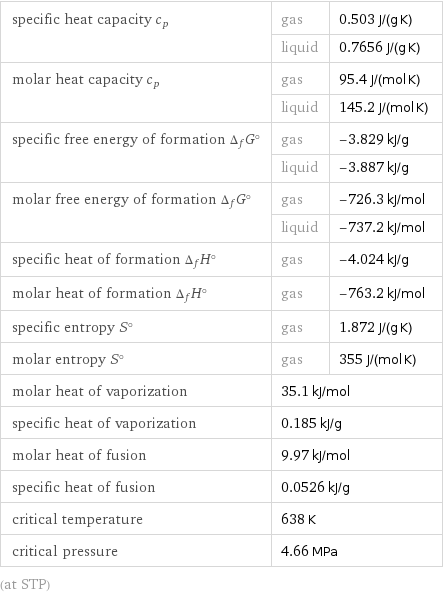
specific heat capacity c_p | gas | 0.503 J/(g K) | liquid | 0.7656 J/(g K) molar heat capacity c_p | gas | 95.4 J/(mol K) | liquid | 145.2 J/(mol K) specific free energy of formation Δ_fG° | gas | -3.829 kJ/g | liquid | -3.887 kJ/g molar free energy of formation Δ_fG° | gas | -726.3 kJ/mol | liquid | -737.2 kJ/mol specific heat of formation Δ_fH° | gas | -4.024 kJ/g molar heat of formation Δ_fH° | gas | -763.2 kJ/mol specific entropy S° | gas | 1.872 J/(g K) molar entropy S° | gas | 355 J/(mol K) molar heat of vaporization | 35.1 kJ/mol | specific heat of vaporization | 0.185 kJ/g | molar heat of fusion | 9.97 kJ/mol | specific heat of fusion | 0.0526 kJ/g | critical temperature | 638 K | critical pressure | 4.66 MPa | (at STP)
Chemical identifiers
(Cl)Cl InChI identifier | InChI=1/4ClH.Ti/h4*1H;/q;;;;+4/p-4/f4Cl.Ti/h4*1h;/q4*-1;m/rCl4Ti/c1-5(2, 3)4 RTECS number | XR1925000 MDL number | MFCD00011267](../image_source/925d134065e43ada1e4145488e1e7d1c.png)
CAS number | 7550-45-0 PubChem CID number | 24193 PubChem SID number | 24852522 SMILES identifier | Cl[Ti](Cl)(Cl)Cl InChI identifier | InChI=1/4ClH.Ti/h4*1H;/q;;;;+4/p-4/f4Cl.Ti/h4*1h;/q4*-1;m/rCl4Ti/c1-5(2, 3)4 RTECS number | XR1925000 MDL number | MFCD00011267
NFPA label

NFPA label
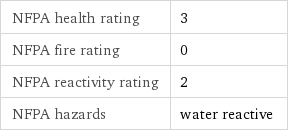
NFPA health rating | 3 NFPA fire rating | 0 NFPA reactivity rating | 2 NFPA hazards | water reactive
Toxicity properties

RTECS classes | other