Input interpretation
sodium chlorate
Chemical names and formulas

formula | NaClO_3 Hill formula | ClNaO_3 name | sodium chlorate alternate names | agrosan | atl acid e | desolet | soda chlorate mass fractions | Cl (chlorine) 33.3% | Na (sodium) 21.6% | O (oxygen) 45.1%
Structure diagram
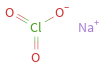
Structure diagram
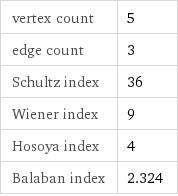
vertex count | 5 edge count | 3 Schultz index | 36 Wiener index | 9 Hosoya index | 4 Balaban index | 2.324
Basic properties

molar mass | 106.4 g/mol phase | liquid (at STP) boiling point | 106 °C density | 1.3 g/cm^3 solubility in water | very soluble
Units

Liquid properties (at STP)
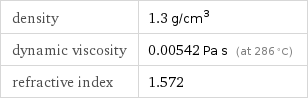
density | 1.3 g/cm^3 dynamic viscosity | 0.00542 Pa s (at 286 °C) refractive index | 1.572
Units

Thermodynamic properties
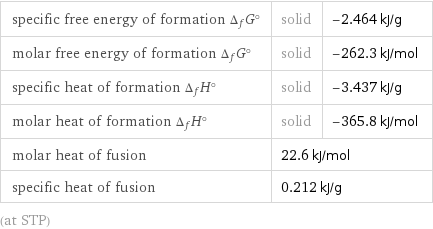
specific free energy of formation Δ_fG° | solid | -2.464 kJ/g molar free energy of formation Δ_fG° | solid | -262.3 kJ/mol specific heat of formation Δ_fH° | solid | -3.437 kJ/g molar heat of formation Δ_fH° | solid | -365.8 kJ/mol molar heat of fusion | 22.6 kJ/mol | specific heat of fusion | 0.212 kJ/g | (at STP)
Chemical identifiers
![CAS number | 7775-09-9 PubChem CID number | 24487 SMILES identifier | [O-]Cl(=O)=O.[Na+] InChI identifier | InChI=1/ClHO3.Na/c2-1(3)4;/h(H, 2, 3, 4);/q;+1/p-1/fClO3.Na/q-1;m EU number | 231-887-4 Gmelin number | 17881 RTECS number | FO0525000](../image_source/720d50447785fafb821c31b146c0fe7a.png)
CAS number | 7775-09-9 PubChem CID number | 24487 SMILES identifier | [O-]Cl(=O)=O.[Na+] InChI identifier | InChI=1/ClHO3.Na/c2-1(3)4;/h(H, 2, 3, 4);/q;+1/p-1/fClO3.Na/q-1;m EU number | 231-887-4 Gmelin number | 17881 RTECS number | FO0525000
NFPA label
NFPA label
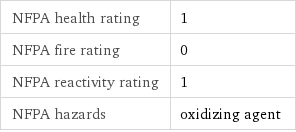
NFPA health rating | 1 NFPA fire rating | 0 NFPA reactivity rating | 1 NFPA hazards | oxidizing agent
Toxicity properties

odor | odorless

RTECS classes | agricultural chemical and pesticide | tumorigen | mutagen | human data | primary irritant
Ion equivalents

Na^+ (sodium cation) | 1 (ClO_3)^- (chlorate anion) | 1