Input interpretation
radium | common compound names
Result
radium sulfate
Chemical names and formulas

formula | RaSO_4 Hill formula | O_4RaS-2 name | radium sulfate mass fractions | O (oxygen) 19.9% | Ra (radium) 70.2% | S (sulfur) 9.95%
Structure diagram
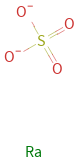
Structure diagram
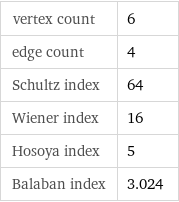
vertex count | 6 edge count | 4 Schultz index | 64 Wiener index | 16 Hosoya index | 5 Balaban index | 3.024
Basic properties

molar mass | 322 g/mol
Units

Thermodynamic properties

specific free energy of formation Δ_fG° | solid | -4.24 kJ/g molar free energy of formation Δ_fG° | solid | -1366 kJ/mol specific heat of formation Δ_fH° | solid | -4.568 kJ/g molar heat of formation Δ_fH° | solid | -1471 kJ/mol (at STP)
Chemical identifiers
![PubChem CID number | 6365199 SMILES identifier | [O-]S(=O)(=O)[O-].[Ra] InChI identifier | InChI=1/H2O4S.Ra/c1-5(2, 3)4;/h(H2, 1, 2, 3, 4);/p-2/fO4S.Ra/q-2;](../image_source/0c884248f01fe64f5ed99f77cfb8a64b.png)
PubChem CID number | 6365199 SMILES identifier | [O-]S(=O)(=O)[O-].[Ra] InChI identifier | InChI=1/H2O4S.Ra/c1-5(2, 3)4;/h(H2, 1, 2, 3, 4);/p-2/fO4S.Ra/q-2;