Input interpretation

vanadium(II) oxide | praseodymium(III) chloride
Chemical names and formulas
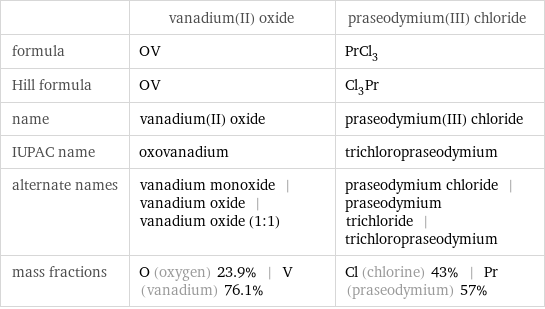
| vanadium(II) oxide | praseodymium(III) chloride formula | OV | PrCl_3 Hill formula | OV | Cl_3Pr name | vanadium(II) oxide | praseodymium(III) chloride IUPAC name | oxovanadium | trichloropraseodymium alternate names | vanadium monoxide | vanadium oxide | vanadium oxide (1:1) | praseodymium chloride | praseodymium trichloride | trichloropraseodymium mass fractions | O (oxygen) 23.9% | V (vanadium) 76.1% | Cl (chlorine) 43% | Pr (praseodymium) 57%
Structure diagrams
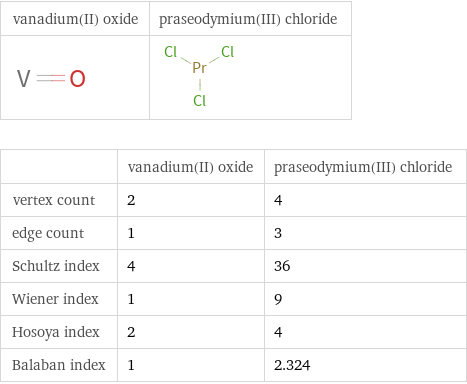
| vanadium(II) oxide | praseodymium(III) chloride vertex count | 2 | 4 edge count | 1 | 3 Schultz index | 4 | 36 Wiener index | 1 | 9 Hosoya index | 2 | 4 Balaban index | 1 | 2.324
3D structure
3D structure
Basic properties
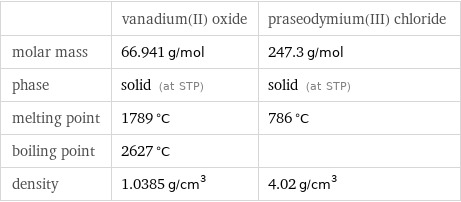
| vanadium(II) oxide | praseodymium(III) chloride molar mass | 66.941 g/mol | 247.3 g/mol phase | solid (at STP) | solid (at STP) melting point | 1789 °C | 786 °C boiling point | 2627 °C | density | 1.0385 g/cm^3 | 4.02 g/cm^3
Units

Thermodynamic properties
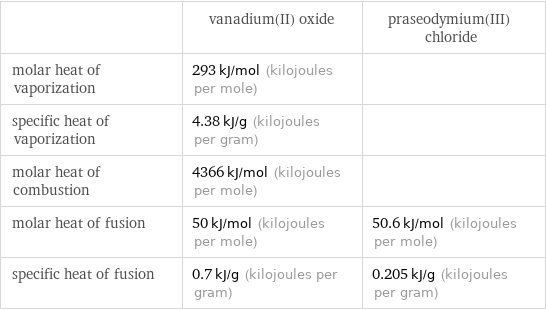
| vanadium(II) oxide | praseodymium(III) chloride molar heat of vaporization | 293 kJ/mol (kilojoules per mole) | specific heat of vaporization | 4.38 kJ/g (kilojoules per gram) | molar heat of combustion | 4366 kJ/mol (kilojoules per mole) | molar heat of fusion | 50 kJ/mol (kilojoules per mole) | 50.6 kJ/mol (kilojoules per mole) specific heat of fusion | 0.7 kJ/g (kilojoules per gram) | 0.205 kJ/g (kilojoules per gram)
Solid properties (at STP)
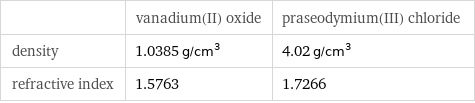
| vanadium(II) oxide | praseodymium(III) chloride density | 1.0385 g/cm^3 | 4.02 g/cm^3 refractive index | 1.5763 | 1.7266
Units

Chemical identifiers
![| vanadium(II) oxide | praseodymium(III) chloride CAS number | 12035-98-2 | 10361-79-2 PubChem CID number | 24411 | 66317 PubChem SID number | | 24857974 SMILES identifier | O=[V] | Cl[Pr](Cl)Cl](../image_source/9a4b6f933fd3e964b209f34585830f43.png)
| vanadium(II) oxide | praseodymium(III) chloride CAS number | 12035-98-2 | 10361-79-2 PubChem CID number | 24411 | 66317 PubChem SID number | | 24857974 SMILES identifier | O=[V] | Cl[Pr](Cl)Cl