Input interpretation
zinc nitrate hydrate
Chemical names and formulas

formula | Zn(NO_3)_2·xH_2O Hill formula | H_12N_2O_12Zn name | zinc nitrate hydrate IUPAC name | zinc dinitrate hexahydrate alternate names | zinc dinitrate hexahydrate mass fractions | H (hydrogen) 4.07% | N (nitrogen) 9.42% | O (oxygen) 64.5% | Zn (zinc) 22%
Structure diagram
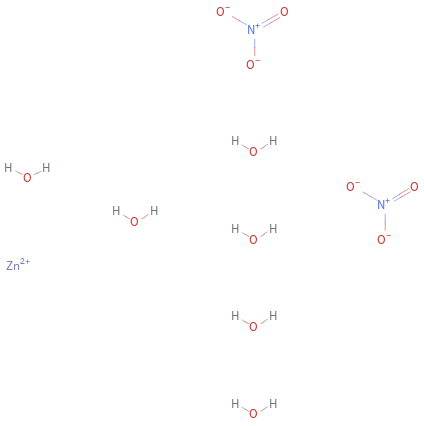
Structure diagram
Basic properties
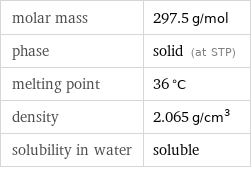
molar mass | 297.5 g/mol phase | solid (at STP) melting point | 36 °C density | 2.065 g/cm^3 solubility in water | soluble
Units

Solid properties (at STP)

density | 2.065 g/cm^3
Units

Chemical identifiers
([O-])[O-].[N+](=O)([O-])[O-].O.O.O.O.O.O.[Zn+2] InChI identifier | InChI=1/2NO3.6H2O.Zn/c2*2-1(3)4;;;;;;;/h;;6*1H2;/q2*-1;;;;;;;+2 RTECS number | ZH4775000 MDL number | MFCD00149889](../image_source/e69468c95562261ee9fcafe47645ce2b.png)
CAS number | 13778-30-8 PubChem CID number | 15865313 PubChem SID number | 24853768 SMILES identifier | [N+](=O)([O-])[O-].[N+](=O)([O-])[O-].O.O.O.O.O.O.[Zn+2] InChI identifier | InChI=1/2NO3.6H2O.Zn/c2*2-1(3)4;;;;;;;/h;;6*1H2;/q2*-1;;;;;;;+2 RTECS number | ZH4775000 MDL number | MFCD00149889
Ion equivalents

Zn^(2+) (zinc cation) | 1 (NO_3)^- (nitrate anion) | 2