Input interpretation
mercuric chloride | ammonium tetrafluoroantimonate
Chemical names and formulas
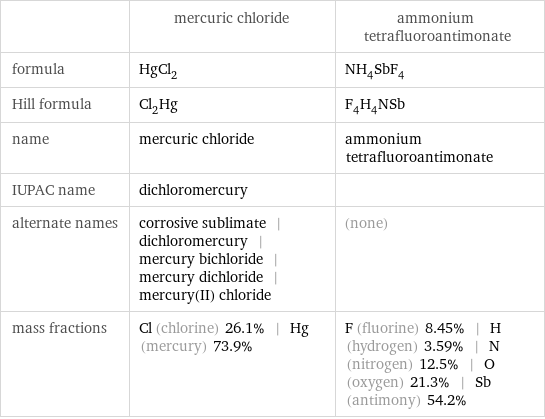
| mercuric chloride | ammonium tetrafluoroantimonate formula | HgCl_2 | NH_4SbF_4 Hill formula | Cl_2Hg | F_4H_4NSb name | mercuric chloride | ammonium tetrafluoroantimonate IUPAC name | dichloromercury | alternate names | corrosive sublimate | dichloromercury | mercury bichloride | mercury dichloride | mercury(II) chloride | (none) mass fractions | Cl (chlorine) 26.1% | Hg (mercury) 73.9% | F (fluorine) 8.45% | H (hydrogen) 3.59% | N (nitrogen) 12.5% | O (oxygen) 21.3% | Sb (antimony) 54.2%
Structure diagrams
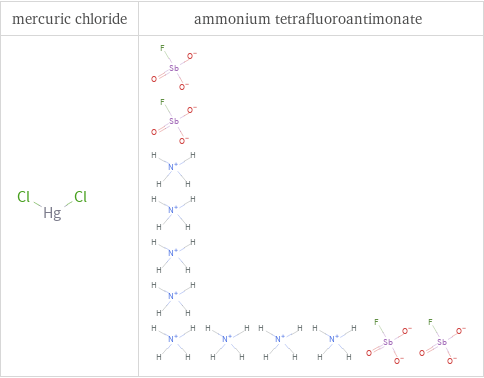
Structure diagrams
Basic properties
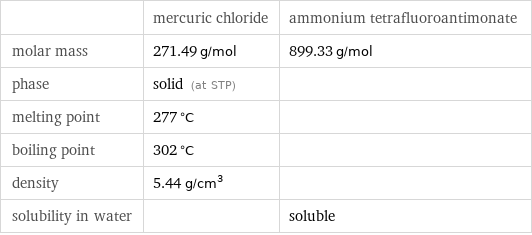
| mercuric chloride | ammonium tetrafluoroantimonate molar mass | 271.49 g/mol | 899.33 g/mol phase | solid (at STP) | melting point | 277 °C | boiling point | 302 °C | density | 5.44 g/cm^3 | solubility in water | | soluble
Units

Thermodynamic properties

| mercuric chloride critical temperature | 973 K (kelvins)
Solid properties
| mercuric chloride density | 5.44 g/cm^3 vapor pressure | 1.3 mmHg
Units
