Input interpretation

cobalt ions | ion class
Structure diagrams

Structure diagrams
Table
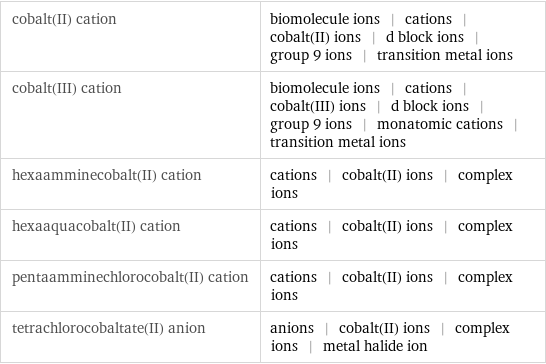
cobalt(II) cation | biomolecule ions | cations | cobalt(II) ions | d block ions | group 9 ions | transition metal ions cobalt(III) cation | biomolecule ions | cations | cobalt(III) ions | d block ions | group 9 ions | monatomic cations | transition metal ions hexaamminecobalt(II) cation | cations | cobalt(II) ions | complex ions hexaaquacobalt(II) cation | cations | cobalt(II) ions | complex ions pentaamminechlorocobalt(II) cation | cations | cobalt(II) ions | complex ions tetrachlorocobaltate(II) anion | anions | cobalt(II) ions | complex ions | metal halide ion
Ionic radii

thermochemical radius | median | 248 pm | lowest | 234 pm (hexaaquacobalt(II) cation) | highest | 319 pm (tetrachlorocobaltate(II) anion)
Units
