Input interpretation
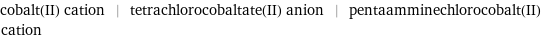
cobalt(II) cation | tetrachlorocobaltate(II) anion | pentaamminechlorocobalt(II) cation
Structure diagrams

Structure diagrams
General properties
![| cobalt(II) cation | tetrachlorocobaltate(II) anion | pentaamminechlorocobalt(II) cation formula | Co^(2+) | ([CoCl_4])^(2-) | ([Co(NH_3)_5Cl])^(2+) net ionic charge | +2 | -2 | +2 alternate names | cobaltous | cobalt(II) | cobalt(2+) | tetrachlorocobaltate(II) | tetrachlorocobaltate(2-) | pentaamminechlorocobalt(II) | pentaamminechlorocobalt(2+)](../image_source/c6a871c9c139a21205284a1f52d1c480.png)
| cobalt(II) cation | tetrachlorocobaltate(II) anion | pentaamminechlorocobalt(II) cation formula | Co^(2+) | ([CoCl_4])^(2-) | ([Co(NH_3)_5Cl])^(2+) net ionic charge | +2 | -2 | +2 alternate names | cobaltous | cobalt(II) | cobalt(2+) | tetrachlorocobaltate(II) | tetrachlorocobaltate(2-) | pentaamminechlorocobalt(II) | pentaamminechlorocobalt(2+)
Ionic radii

| cobalt(II) cation | tetrachlorocobaltate(II) anion | pentaamminechlorocobalt(II) cation 4-coordinate ionic radius | 56 pm | | 6-coordinate ionic radius | 65 pm | | 8-coordinate ionic radius | 90 pm | | thermochemical radius | | 319 pm | 236 pm
Units

Other properties

| cobalt(II) cation | tetrachlorocobaltate(II) anion | pentaamminechlorocobalt(II) cation ion class | biomolecule ions | cations | cobalt(II) ions | d block ions | group 9 ions | transition metal ions | anions | cobalt(II) ions | complex ions | metal halide ion | cations | cobalt(II) ions | complex ions common sources of ion | cobalt dithiocyanate (1 eq) | cobalt(II) sulfate hydrate (1 eq) | cobalt(II) stearate (1 eq) | cobalt phthalocyanine (1 eq) | cobalt(II) phosphate hydrate (3 eq) | cobaltous nitrate hexahydrate (1 eq) | cobalt cyanide dihydrate (1 eq) | cobalt(II) carbonate hydrate (1 eq) | cobalt(II) 2, 3-naphthalocyanine (1 eq) | |