Input interpretation

greenhouse gases | molar mass
Summary
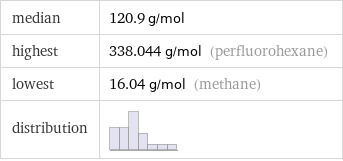
median | 120.9 g/mol highest | 338.044 g/mol (perfluorohexane) lowest | 16.04 g/mol (methane) distribution |
Units

Distribution plots
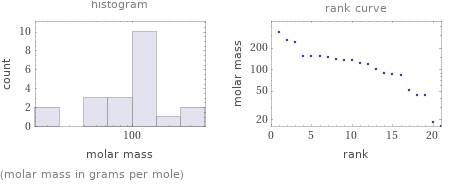
(molar mass in grams per mole)
Molar mass rankings
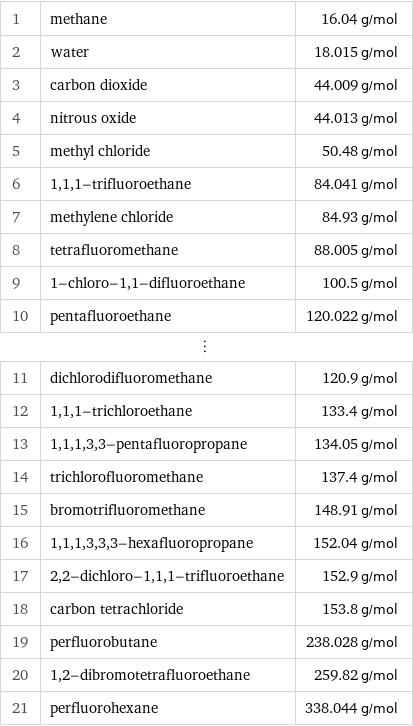
1 | methane | 16.04 g/mol 2 | water | 18.015 g/mol 3 | carbon dioxide | 44.009 g/mol 4 | nitrous oxide | 44.013 g/mol 5 | methyl chloride | 50.48 g/mol 6 | 1, 1, 1-trifluoroethane | 84.041 g/mol 7 | methylene chloride | 84.93 g/mol 8 | tetrafluoromethane | 88.005 g/mol 9 | 1-chloro-1, 1-difluoroethane | 100.5 g/mol 10 | pentafluoroethane | 120.022 g/mol ⋮ | | 11 | dichlorodifluoromethane | 120.9 g/mol 12 | 1, 1, 1-trichloroethane | 133.4 g/mol 13 | 1, 1, 1, 3, 3-pentafluoropropane | 134.05 g/mol 14 | trichlorofluoromethane | 137.4 g/mol 15 | bromotrifluoromethane | 148.91 g/mol 16 | 1, 1, 1, 3, 3, 3-hexafluoropropane | 152.04 g/mol 17 | 2, 2-dichloro-1, 1, 1-trifluoroethane | 152.9 g/mol 18 | carbon tetrachloride | 153.8 g/mol 19 | perfluorobutane | 238.028 g/mol 20 | 1, 2-dibromotetrafluoroethane | 259.82 g/mol 21 | perfluorohexane | 338.044 g/mol
Unit conversion for median molar mass 120.9 g/mol
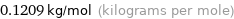
0.1209 kg/mol (kilograms per mole)
Comparisons for median molar mass 120.9 g/mol
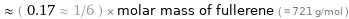
≈ ( 0.17 ≈ 1/6 ) × molar mass of fullerene ( ≈ 721 g/mol )
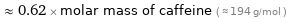
≈ 0.62 × molar mass of caffeine ( ≈ 194 g/mol )
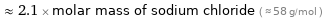
≈ 2.1 × molar mass of sodium chloride ( ≈ 58 g/mol )
Corresponding quantities
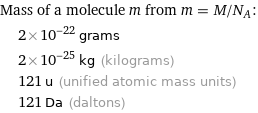
Mass of a molecule m from m = M/N_A: | 2×10^-22 grams | 2×10^-25 kg (kilograms) | 121 u (unified atomic mass units) | 121 Da (daltons)
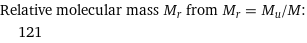
Relative molecular mass M_r from M_r = M_u/M: | 121