Input interpretation
dactinomycin
Chemical names and formulas
![formula | C_62H_86N_12O_16 name | dactinomycin IUPAC name | 2-amino-4, 6-dimethyl-3-oxo-N, N'-bis[(3R, 6S, 7R, 10S, 16S)-7, 11, 14-trimethyl-2, 5, 9, 12, 15-pentaoxo-3, 10-dipropan-2-yl-8-oxa-1, 4, 11, 14-tetrazabicyclo[14.3.0]nonadec-6-yl]phenoxazine-1, 9-dicarboxamide alternate names | actinomycin c1 | actinomycin d | actinomycin i mass fractions | C (carbon) 59.3% | H (hydrogen) 6.91% | N (nitrogen) 13.4% | O (oxygen) 20.4%](../image_source/325592e9402aebc945e1e6e855d70db9.png)
formula | C_62H_86N_12O_16 name | dactinomycin IUPAC name | 2-amino-4, 6-dimethyl-3-oxo-N, N'-bis[(3R, 6S, 7R, 10S, 16S)-7, 11, 14-trimethyl-2, 5, 9, 12, 15-pentaoxo-3, 10-dipropan-2-yl-8-oxa-1, 4, 11, 14-tetrazabicyclo[14.3.0]nonadec-6-yl]phenoxazine-1, 9-dicarboxamide alternate names | actinomycin c1 | actinomycin d | actinomycin i mass fractions | C (carbon) 59.3% | H (hydrogen) 6.91% | N (nitrogen) 13.4% | O (oxygen) 20.4%
Lewis structure
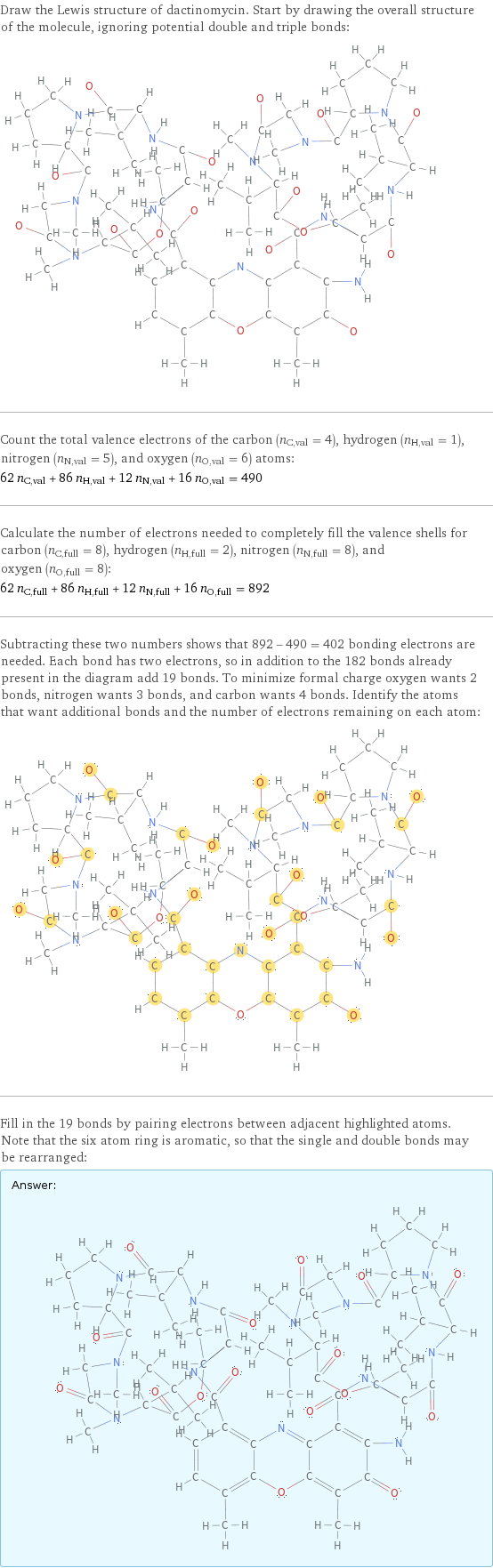
Draw the Lewis structure of dactinomycin. Start by drawing the overall structure of the molecule, ignoring potential double and triple bonds: Count the total valence electrons of the carbon (n_C, val = 4), hydrogen (n_H, val = 1), nitrogen (n_N, val = 5), and oxygen (n_O, val = 6) atoms: 62 n_C, val + 86 n_H, val + 12 n_N, val + 16 n_O, val = 490 Calculate the number of electrons needed to completely fill the valence shells for carbon (n_C, full = 8), hydrogen (n_H, full = 2), nitrogen (n_N, full = 8), and oxygen (n_O, full = 8): 62 n_C, full + 86 n_H, full + 12 n_N, full + 16 n_O, full = 892 Subtracting these two numbers shows that 892 - 490 = 402 bonding electrons are needed. Each bond has two electrons, so in addition to the 182 bonds already present in the diagram add 19 bonds. To minimize formal charge oxygen wants 2 bonds, nitrogen wants 3 bonds, and carbon wants 4 bonds. Identify the atoms that want additional bonds and the number of electrons remaining on each atom: Fill in the 19 bonds by pairing electrons between adjacent highlighted atoms. Note that the six atom ring is aromatic, so that the single and double bonds may be rearranged: Answer: | |
3D structure
3D structure
Basic properties
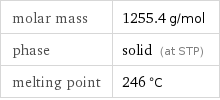
molar mass | 1255.4 g/mol phase | solid (at STP) melting point | 246 °C
Units

Hydrophobicity and permeability properties

experimental LogP hydrophobicity | 1.6 predicted LogP hydrophobicity | 2.76 predicted LogS | -4.8
Basic drug properties

approval status | approved | small molecule drug categories | anti-bacterial agent | antibiotic | antineoplastic antibiotic | antineoplastic agent | nucleic acid synthesis inhibitor | protein synthesis inhibitor dosage forms | intravenous: powder, for solution

brand names | ACT | ACT D | ACTINOMYCIN D | ACTO-D | actactinomycin a Iv | actinomycin 11 cosmegen | actinomycin 7 | actinomycin aiv | actinomycin C1 | actinomycin I | actinomycin I1 | actinomycin Iv | actinomycin X 1 | actinomycindioic D acid, dilactone | chounghwamycin B | cosmegen | dactinomycin D | dilactone actinomycin D acid | dilactone actinomycindioic D acid | HBF 386 meractinomycin | lyovac cosmegen | meractinomycin | oncostatin K | oxamide
Chemical identifiers

CAS number | 50-76-0 Beilstein number | 605235 PubChem CID number | 457193 PubChem SID number | 8990 SMILES identifier | CC1C(C(=O)NC(C(=O)N2CCCC2C(=O)N(CC(=O)N(C(C(=O)O1)C(C)C)C)C)C(C)C)NC(=O)C3=C4C(=C(C=C3)C)OC5=C(C(=O)C(=C(C5=N4)C(=O)NC6C(OC(=O)C(N(C(=O)CN(C(=O)C7CCCN7C(=O)C(NC6=O)C(C)C)C)C)C(C)C)C)N)C InChI identifier | InChI=1/C62H86N12O16/c1-27(2)42-59(84)73-23-17-19-36(73)57(82)69(13)25-38(75)71(15)48(29(5)6)61(86)88-33(11)44(55(80)65-42)67-53(78)35-22-21-31(9)51-46(35)64-47-40(41(63)50(77)32(10)52(47)90-51)54(79)68-45-34(12)89-62(87)49(30(7)8)72(16)39(76)26-70(14)58(83)37-20-18-24-74(37)60(85)43(28(3)4)66-56(45)81/h21-22, 27-30, 33-34, 36-37, 42-45, 48-49H, 17-20, 23-26, 63H2, 1-16H3, (H, 65, 80)(H, 66, 81)(H, 67, 78)(H, 68, 79)/t33-, 34-, 36+, 37+, 42+, 43+, 44+, 45+, 48+, 49-/m1/s1/f/h65-68H InChI key | RJURFGZVJUQBHK-YKMFUUCJCA EU number | 200-063-6 RTECS number | AU1575000 NSC number | 3053
NFPA label

NFPA label

NFPA health rating | 4 NFPA fire rating | 0 NFPA reactivity rating | 0
Safety properties

flash point | 241 °C
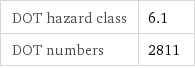
DOT hazard class | 6.1 DOT numbers | 2811
Toxicity properties

RTECS classes | tumorigen | drug | mutagen | reproductive effector | human data | natural product | primary irritant