Input interpretation
potassium hydride
Chemical names and formulas

formula | KH Hill formula | HK name | potassium hydride alternate names | potassium hydride (1:1) | potassium monohydride mass fractions | H (hydrogen) 2.51% | K (potassium) 97.5%
Structure diagram
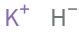
Structure diagram
Basic properties
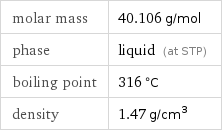
molar mass | 40.106 g/mol phase | liquid (at STP) boiling point | 316 °C density | 1.47 g/cm^3
Units

Liquid properties (at STP)

density | 1.47 g/cm^3
Units

Thermodynamic properties

specific heat of formation Δ_fH° | solid | -1.439 kJ/g molar heat of formation Δ_fH° | solid | -57.7 kJ/mol molar heat of fusion | 21 kJ/mol | specific heat of fusion | 0.52 kJ/g | (at STP)
Chemical identifiers
![CAS number | 7693-26-7 PubChem CID number | 82127 PubChem SID number | 24398127 SMILES identifier | [H-].[K+] InChI identifier | InChI=1/K.H/q+1;-1 MDL number | MFCD00011357](../image_source/052fb16af85ac0b57b5f670c0da0ca4b.png)
CAS number | 7693-26-7 PubChem CID number | 82127 PubChem SID number | 24398127 SMILES identifier | [H-].[K+] InChI identifier | InChI=1/K.H/q+1;-1 MDL number | MFCD00011357
NFPA label

NFPA label

NFPA hazards | water reactive