Input interpretation

platinum metals | molar heat of vaporization
Summary
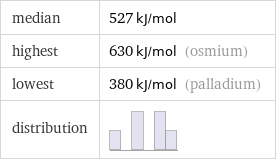
median | 527 kJ/mol highest | 630 kJ/mol (osmium) lowest | 380 kJ/mol (palladium) distribution |
Units

Ranked values
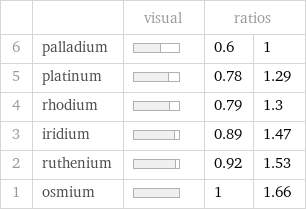
| | visual | ratios | 6 | palladium | | 0.6 | 1 5 | platinum | | 0.78 | 1.29 4 | rhodium | | 0.79 | 1.3 3 | iridium | | 0.89 | 1.47 2 | ruthenium | | 0.92 | 1.53 1 | osmium | | 1 | 1.66
Molar heat of vaporization rankings
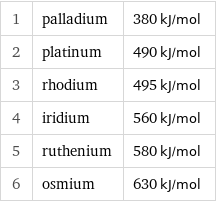
1 | palladium | 380 kJ/mol 2 | platinum | 490 kJ/mol 3 | rhodium | 495 kJ/mol 4 | iridium | 560 kJ/mol 5 | ruthenium | 580 kJ/mol 6 | osmium | 630 kJ/mol
Unit conversions for median molar heat of vaporization 527 kJ/mol
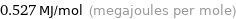
0.527 MJ/mol (megajoules per mole)
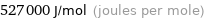
527000 J/mol (joules per mole)
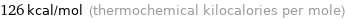
126 kcal/mol (thermochemical kilocalories per mole)
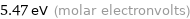
5.47 eV (molar electronvolts)