Input interpretation
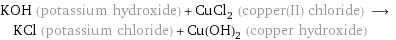
KOH (potassium hydroxide) + CuCl_2 (copper(II) chloride) ⟶ KCl (potassium chloride) + Cu(OH)_2 (copper hydroxide)
Balanced equation

Balance the chemical equation algebraically: KOH + CuCl_2 ⟶ KCl + Cu(OH)_2 Add stoichiometric coefficients, c_i, to the reactants and products: c_1 KOH + c_2 CuCl_2 ⟶ c_3 KCl + c_4 Cu(OH)_2 Set the number of atoms in the reactants equal to the number of atoms in the products for H, K, O, Cl and Cu: H: | c_1 = 2 c_4 K: | c_1 = c_3 O: | c_1 = 2 c_4 Cl: | 2 c_2 = c_3 Cu: | c_2 = c_4 Since the coefficients are relative quantities and underdetermined, choose a coefficient to set arbitrarily. To keep the coefficients small, the arbitrary value is ordinarily one. For instance, set c_2 = 1 and solve the system of equations for the remaining coefficients: c_1 = 2 c_2 = 1 c_3 = 2 c_4 = 1 Substitute the coefficients into the chemical reaction to obtain the balanced equation: Answer: | | 2 KOH + CuCl_2 ⟶ 2 KCl + Cu(OH)_2
Structures
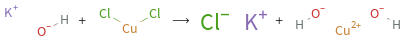
+ ⟶ +
Names
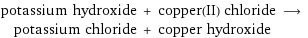
potassium hydroxide + copper(II) chloride ⟶ potassium chloride + copper hydroxide
Equilibrium constant
![K_c = ([KCl]^2 [Cu(OH)2])/([KOH]^2 [CuCl2])](../image_source/5178099dabd65c00c18b4c77ffa79e28.png)
K_c = ([KCl]^2 [Cu(OH)2])/([KOH]^2 [CuCl2])
Rate of reaction
![rate = -1/2 (Δ[KOH])/(Δt) = -(Δ[CuCl2])/(Δt) = 1/2 (Δ[KCl])/(Δt) = (Δ[Cu(OH)2])/(Δt) (assuming constant volume and no accumulation of intermediates or side products)](../image_source/312ba26e5de1ff6fd8f3e00d97ec57ee.png)
rate = -1/2 (Δ[KOH])/(Δt) = -(Δ[CuCl2])/(Δt) = 1/2 (Δ[KCl])/(Δt) = (Δ[Cu(OH)2])/(Δt) (assuming constant volume and no accumulation of intermediates or side products)
Chemical names and formulas
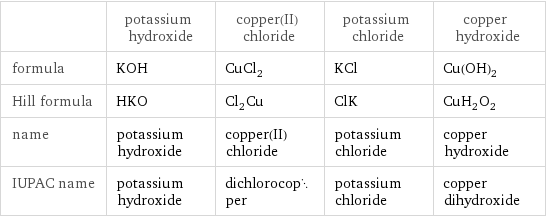
| potassium hydroxide | copper(II) chloride | potassium chloride | copper hydroxide formula | KOH | CuCl_2 | KCl | Cu(OH)_2 Hill formula | HKO | Cl_2Cu | ClK | CuH_2O_2 name | potassium hydroxide | copper(II) chloride | potassium chloride | copper hydroxide IUPAC name | potassium hydroxide | dichlorocopper | potassium chloride | copper dihydroxide
Substance properties
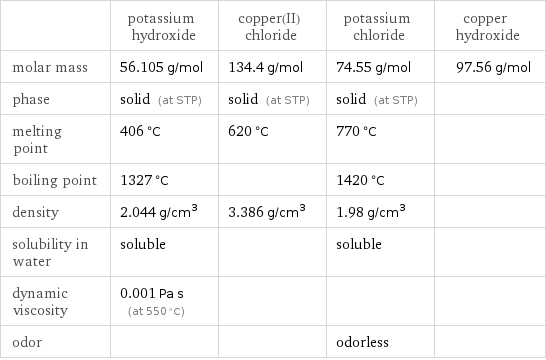
| potassium hydroxide | copper(II) chloride | potassium chloride | copper hydroxide molar mass | 56.105 g/mol | 134.4 g/mol | 74.55 g/mol | 97.56 g/mol phase | solid (at STP) | solid (at STP) | solid (at STP) | melting point | 406 °C | 620 °C | 770 °C | boiling point | 1327 °C | | 1420 °C | density | 2.044 g/cm^3 | 3.386 g/cm^3 | 1.98 g/cm^3 | solubility in water | soluble | | soluble | dynamic viscosity | 0.001 Pa s (at 550 °C) | | | odor | | | odorless |
Units
