Input interpretation
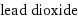
lead dioxide
Chemical names and formulas

formula | PbO_2 Hill formula | O_2Pb name | lead dioxide alternate names | diketolead | dioxolead | lead brown | lead(IV) oxide | lead oxide | lead peroxide | lead(su)peroxide mass fractions | O (oxygen) 13.4% | Pb (lead) 86.6%
Structure diagram
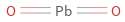
Structure diagram
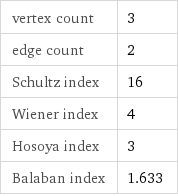
vertex count | 3 edge count | 2 Schultz index | 16 Wiener index | 4 Hosoya index | 3 Balaban index | 1.633
Basic properties
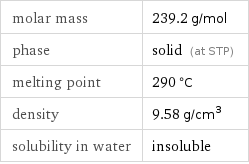
molar mass | 239.2 g/mol phase | solid (at STP) melting point | 290 °C density | 9.58 g/cm^3 solubility in water | insoluble
Units

Solid properties (at STP)

density | 9.58 g/cm^3
Units

Thermodynamic properties
specific free energy of formation Δ_fG° | solid | -0.9085 kJ/g molar free energy of formation Δ_fG° | solid | -217.3 kJ/mol specific heat of formation Δ_fH° | solid | -1.16 kJ/g molar heat of formation Δ_fH° | solid | -277.4 kJ/mol specific entropy S° | solid | 0.2885 J/(g K) molar entropy S° | solid | 69 J/(mol K) (at STP)
Chemical identifiers
![CAS number | 1309-60-0 SMILES identifier | O=[Pb]=O RTECS number | OG0700000 MDL number | MFCD00011165](../image_source/09ee99cf481529a236a2bc4e4f11bd21.png)
CAS number | 1309-60-0 SMILES identifier | O=[Pb]=O RTECS number | OG0700000 MDL number | MFCD00011165
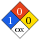
NFPA label
NFPA label
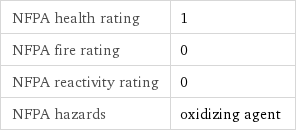
NFPA health rating | 1 NFPA fire rating | 0 NFPA reactivity rating | 0 NFPA hazards | oxidizing agent
Safety properties

flash point | 290 °C
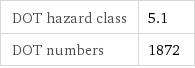
DOT hazard class | 5.1 DOT numbers | 1872
Toxicity properties

RTECS classes | other