Input interpretation
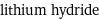
lithium hydride
Chemical names and formulas

formula | LiH Hill formula | HLi name | lithium hydride alternate names | hydridolithium | hydrure de lithium | lithium monohydride mass fractions | H (hydrogen) 12.7% | Li (lithium) 87.3%
Structure diagram

Structure diagram
Basic properties
molar mass | 7.95 g/mol phase | solid (at STP) melting point | 680 °C density | 0.82 g/cm^3 solubility in water | reacts
Units

Solid properties (at STP)
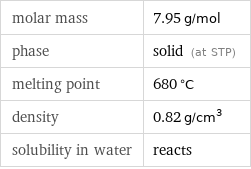
density | 0.82 g/cm^3 vapor pressure | 10 mmHg (at 720 °C)
Units

Thermodynamic properties
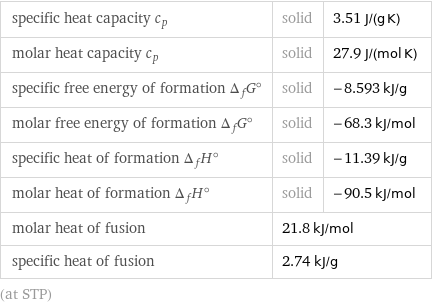
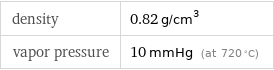
specific heat capacity c_p | solid | 3.51 J/(g K) molar heat capacity c_p | solid | 27.9 J/(mol K) specific free energy of formation Δ_fG° | solid | -8.593 kJ/g molar free energy of formation Δ_fG° | solid | -68.3 kJ/mol specific heat of formation Δ_fH° | solid | -11.39 kJ/g molar heat of formation Δ_fH° | solid | -90.5 kJ/mol molar heat of fusion | 21.8 kJ/mol | specific heat of fusion | 2.74 kJ/g | (at STP)
Chemical identifiers
![CAS number | 7580-67-8 PubChem CID number | 62714 PubChem SID number | 8146271 SMILES identifier | [H-].[Li+] InChI identifier | InChI=1/Li.H/q+1;-1 RTECS number | OJ6300000 MDL number | MFCD00011074](../image_source/7bae936e1934afecd532d614b7eeec00.png)
CAS number | 7580-67-8 PubChem CID number | 62714 PubChem SID number | 8146271 SMILES identifier | [H-].[Li+] InChI identifier | InChI=1/Li.H/q+1;-1 RTECS number | OJ6300000 MDL number | MFCD00011074
NFPA label

NFPA label
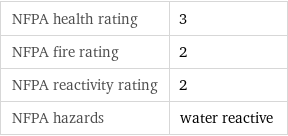
NFPA health rating | 3 NFPA fire rating | 2 NFPA reactivity rating | 2 NFPA hazards | water reactive
Safety properties

autoignition point | 200 °C
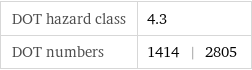
DOT hazard class | 4.3 DOT numbers | 1414 | 2805
Toxicity properties

long-term exposure limit | 0.025 mg/m^3 (over 8 hours) RTECS classes | human data
Units
