Input interpretation
thallium(III) cation | iron(III) cation | oxide anion
Structure diagrams

Structure diagrams
General properties

| thallium(III) cation | iron(III) cation | oxide anion formula | Tl^(3+) | Fe^(3+) | O^(2-) net ionic charge | +3 | +3 | -2 alternate names | thallium(III) | thallium(3+) | ferric | iron(III) | iron(3+) | oxide | oxide(2-)
Ionic radii
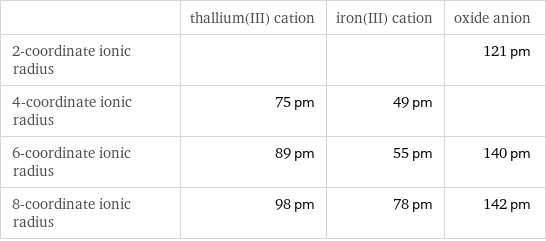
| thallium(III) cation | iron(III) cation | oxide anion 2-coordinate ionic radius | | | 121 pm 4-coordinate ionic radius | 75 pm | 49 pm | 6-coordinate ionic radius | 89 pm | 55 pm | 140 pm 8-coordinate ionic radius | 98 pm | 78 pm | 142 pm
Units

Other properties
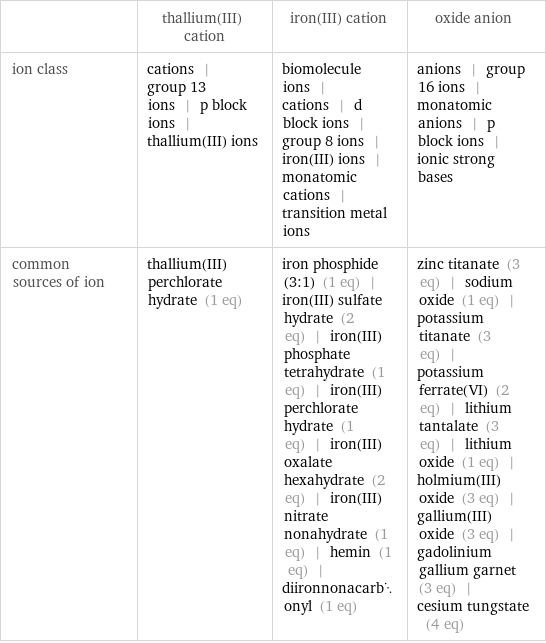
| thallium(III) cation | iron(III) cation | oxide anion ion class | cations | group 13 ions | p block ions | thallium(III) ions | biomolecule ions | cations | d block ions | group 8 ions | iron(III) ions | monatomic cations | transition metal ions | anions | group 16 ions | monatomic anions | p block ions | ionic strong bases common sources of ion | thallium(III) perchlorate hydrate (1 eq) | iron phosphide (3:1) (1 eq) | iron(III) sulfate hydrate (2 eq) | iron(III) phosphate tetrahydrate (1 eq) | iron(III) perchlorate hydrate (1 eq) | iron(III) oxalate hexahydrate (2 eq) | iron(III) nitrate nonahydrate (1 eq) | hemin (1 eq) | diironnonacarbonyl (1 eq) | zinc titanate (3 eq) | sodium oxide (1 eq) | potassium titanate (3 eq) | potassium ferrate(VI) (2 eq) | lithium tantalate (3 eq) | lithium oxide (1 eq) | holmium(III) oxide (3 eq) | gallium(III) oxide (3 eq) | gadolinium gallium garnet (3 eq) | cesium tungstate (4 eq)