Input interpretation
lead tetrachloride | barium trifluoromethanesulfonate
Chemical names and formulas

| lead tetrachloride | barium trifluoromethanesulfonate formula | Cl_4Pb | (CF_3SO_3)_2Ba Hill formula | Cl_4Pb | BaC_2F_6O_6S_2 name | lead tetrachloride | barium trifluoromethanesulfonate IUPAC name | tetrachloroplumbane | alternate names | lead chloride | lead(IV) chloride | plumbane, tetrachloro- | tetrachloro-plumbane | barium triflate | barium; trifluoromethanesulfonic acid | barium trifluoromethanesulphonate | methanesulfonic acid, trifluoro-, barium salt | trifluoromethanesulfonic acid barium salt mass fractions | Cl (chlorine) 40.6% | Pb (lead) 59.4% | Ba (barium) 31.5% | C (carbon) 5.52% | F (fluorine) 26.2% | O (oxygen) 22% | S (sulfur) 14.7%
Structure diagrams
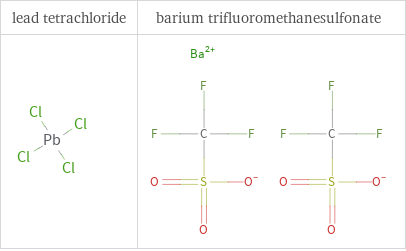
Structure diagrams
Basic properties

| lead tetrachloride | barium trifluoromethanesulfonate molar mass | 349 g/mol | 435.45 g/mol phase | liquid (at STP) | solid (at STP) melting point | -15 °C | 300 °C density | 3.18 g/cm^3 |
Units

Liquid properties
| lead tetrachloride density | 3.18 g/cm^3
Units
