Input interpretation
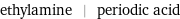
ethylamine | periodic acid
Chemical names and formulas
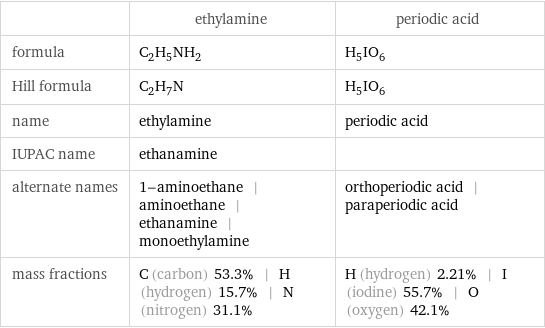
| ethylamine | periodic acid formula | C_2H_5NH_2 | H_5IO_6 Hill formula | C_2H_7N | H_5IO_6 name | ethylamine | periodic acid IUPAC name | ethanamine | alternate names | 1-aminoethane | aminoethane | ethanamine | monoethylamine | orthoperiodic acid | paraperiodic acid mass fractions | C (carbon) 53.3% | H (hydrogen) 15.7% | N (nitrogen) 31.1% | H (hydrogen) 2.21% | I (iodine) 55.7% | O (oxygen) 42.1%
Structure diagrams
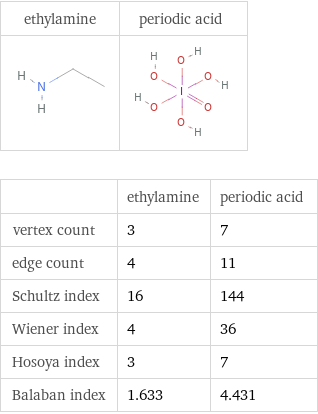
| ethylamine | periodic acid vertex count | 3 | 7 edge count | 4 | 11 Schultz index | 16 | 144 Wiener index | 4 | 36 Hosoya index | 3 | 7 Balaban index | 1.633 | 4.431
3D structure
3D structure
Basic properties
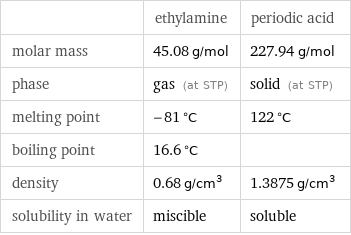
| ethylamine | periodic acid molar mass | 45.08 g/mol | 227.94 g/mol phase | gas (at STP) | solid (at STP) melting point | -81 °C | 122 °C boiling point | 16.6 °C | density | 0.68 g/cm^3 | 1.3875 g/cm^3 solubility in water | miscible | soluble
Units

Gas properties
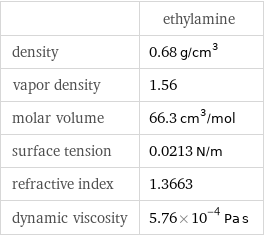
| ethylamine density | 0.68 g/cm^3 vapor density | 1.56 molar volume | 66.3 cm^3/mol surface tension | 0.0213 N/m refractive index | 1.3663 dynamic viscosity | 5.76×10^-4 Pa s
Units

Thermodynamic properties
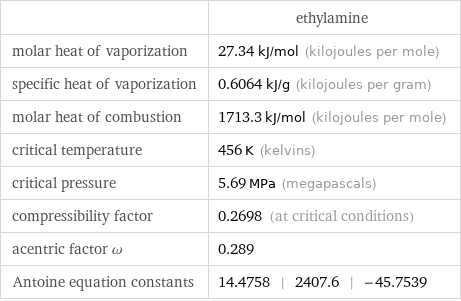
| ethylamine molar heat of vaporization | 27.34 kJ/mol (kilojoules per mole) specific heat of vaporization | 0.6064 kJ/g (kilojoules per gram) molar heat of combustion | 1713.3 kJ/mol (kilojoules per mole) critical temperature | 456 K (kelvins) critical pressure | 5.69 MPa (megapascals) compressibility factor | 0.2698 (at critical conditions) acentric factor ω | 0.289 Antoine equation constants | 14.4758 | 2407.6 | -45.7539