Input interpretation
1 cm^3 of diborane
Basic properties for 1 cm^3

mass | 1.13 mg (milligrams) 0.00113 grams 1.13×10^-6 kg (kilograms) 0.0175 grains molar amount | 40.9 µmol (micromoles) 0.0409 mmol (millimoles) 4.09×10^-5 mol (moles) volume | 1 mL (milliliter) 1000 µL (microliters) 0.001 L (liters) 1×10^-6 m^3 (cubic meters) 0.203 tsp (teaspoons) 0.00106 quarts equivalents | 4.09×10^-5 eq (equivalents) (at STP)
Corresponding quantities

sphere radius | 6.204 mm (millimeters) side of a cube | 10 mm (millimeters)
Thermodynamic properties for 1 cm^3

heat capacity C_p | gaseous | 0.002318 J/K free energy of formation Δ_fG° | gaseous | 0.003581 kJ | gaseous | 0.03164 kJ heat of formation Δ_fH° | gaseous | 0.001488 kJ
Units
Energy vs. temperature for 1 cm^3
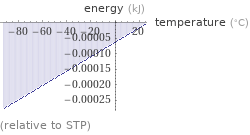
(relative to STP)
Units

Phase change energies for 1 cm^3 from 25 °C
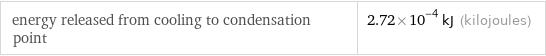
energy released from cooling to condensation point | 2.72×10^-4 kJ (kilojoules)
Mass composition for 1 cm^3
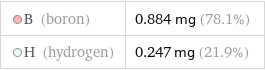
B (boron) | 0.884 mg (78.1%) H (hydrogen) | 0.247 mg (21.9%)
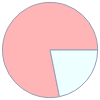
Mass composition for 1 cm^3
Structure diagram
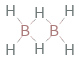
Structure diagram

| bond counts | bond energies | 8 bonds | 389 kJ/mol
Chemical names and formulas

formula | B_2H_6 Hill formula | B_2H_6 name | diborane IUPAC name | diborane
Substance properties
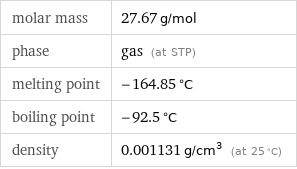
molar mass | 27.67 g/mol phase | gas (at STP) melting point | -164.85 °C boiling point | -92.5 °C density | 0.001131 g/cm^3 (at 25 °C)
Units
