Input interpretation

sodium bromide | molar volume
Result

32 cm^3/mol (cubic centimeters per mole)
Unit conversions
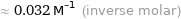
≈ 0.032 M^(-1) (inverse molar)
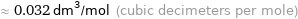
≈ 0.032 dm^3/mol (cubic decimeters per mole)
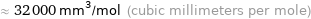
≈ 32000 mm^3/mol (cubic millimeters per mole)
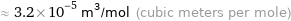
≈ 3.2×10^-5 m^3/mol (cubic meters per mole)
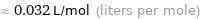
≈ 0.032 L/mol (liters per mole)
Comparison as molar volume
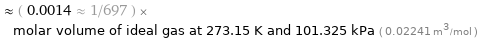
≈ ( 0.0014 ≈ 1/697 ) × molar volume of ideal gas at 273.15 K and 101.325 kPa ( 0.02241 m^3/mol )