Input interpretation
sodium fluoride
Chemical names and formulas

formula | NaF Hill formula | FNa name | sodium fluoride alternate names | antibulit | chemifluor | floridine | sodium monofluoride mass fractions | F (fluorine) 45.2% | Na (sodium) 54.8%
Structure diagram

Structure diagram
Basic properties
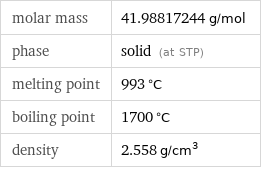
molar mass | 41.98817244 g/mol phase | solid (at STP) melting point | 993 °C boiling point | 1700 °C density | 2.558 g/cm^3
Units

Solid properties (at STP)

density | 2.558 g/cm^3 vapor pressure | 1.4 mmHg
Units

Thermodynamic properties
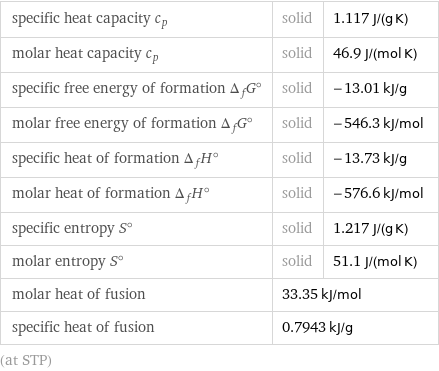
specific heat capacity c_p | solid | 1.117 J/(g K) molar heat capacity c_p | solid | 46.9 J/(mol K) specific free energy of formation Δ_fG° | solid | -13.01 kJ/g molar free energy of formation Δ_fG° | solid | -546.3 kJ/mol specific heat of formation Δ_fH° | solid | -13.73 kJ/g molar heat of formation Δ_fH° | solid | -576.6 kJ/mol specific entropy S° | solid | 1.217 J/(g K) molar entropy S° | solid | 51.1 J/(mol K) molar heat of fusion | 33.35 kJ/mol | specific heat of fusion | 0.7943 kJ/g | (at STP)
Chemical identifiers
![CAS number | 7681-49-4 PubChem CID number | 5235 PubChem SID number | 24852884 SMILES identifier | [F-].[Na+] InChI identifier | InChI=1/FH.Na/h1H;/q;+1/p-1/fF.Na/h1h;/q-1;m RTECS number | WB0350000 MDL number | MFCD00003524](../image_source/fedc3f14e52c6a9cf2747202990a291a.png)
CAS number | 7681-49-4 PubChem CID number | 5235 PubChem SID number | 24852884 SMILES identifier | [F-].[Na+] InChI identifier | InChI=1/FH.Na/h1H;/q;+1/p-1/fF.Na/h1h;/q-1;m RTECS number | WB0350000 MDL number | MFCD00003524
NFPA label

NFPA label

NFPA health rating | 3 NFPA fire rating | 0 NFPA reactivity rating | 0
Toxicity properties

odor | odorless lethal dosage | 52 mg/kg (oral dose for rats)
Units

Ion equivalents

Na^+ (sodium cation) | 1 F^- (fluoride anion) | 1