Input interpretation

copper(II) silicate dihydrate | vanadium carbonyl
Chemical names and formulas
| copper(II) silicate dihydrate | vanadium carbonyl formula | CuSiO_3·2H_2O | V(CO)_6 Hill formula | CuH_4O_5Si | C_6O_6V_1 name | copper(II) silicate dihydrate | vanadium carbonyl mass fractions | Cu (copper) 49.8% | H (hydrogen) 1.58% | O (oxygen) 37.6% | Si (silicon) 11% | C (carbon) 32.9% | O (oxygen) 43.8% | V (vanadium) 23.3%
Structure diagrams
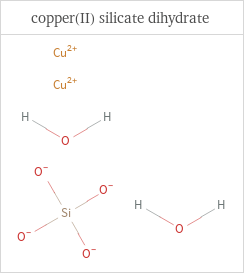
Structure diagrams
Basic properties

| copper(II) silicate dihydrate | vanadium carbonyl molar mass | 255.2 g/mol | 219 g/mol
Units
