Input interpretation

halogens | specific heat at STP
Summary
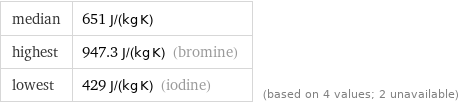
median | 651 J/(kg K) highest | 947.3 J/(kg K) (bromine) lowest | 429 J/(kg K) (iodine) | (based on 4 values; 2 unavailable)
Entities with missing values
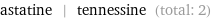
astatine | tennessine (total: 2)
Ranked values
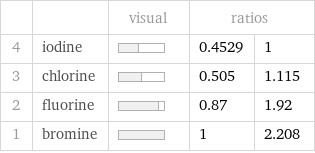
| | visual | ratios | 4 | iodine | | 0.4529 | 1 3 | chlorine | | 0.505 | 1.115 2 | fluorine | | 0.87 | 1.92 1 | bromine | | 1 | 2.208
Specific heat at STP rankings
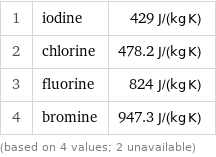
1 | iodine | 429 J/(kg K) 2 | chlorine | 478.2 J/(kg K) 3 | fluorine | 824 J/(kg K) 4 | bromine | 947.3 J/(kg K) (based on 4 values; 2 unavailable)
Unit conversions for median specific heat at STP 651 J/(kg K)
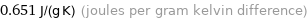
0.651 J/(g K) (joules per gram kelvin difference)
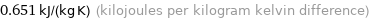
0.651 kJ/(kg K) (kilojoules per kilogram kelvin difference)
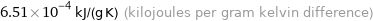
6.51×10^-4 kJ/(g K) (kilojoules per gram kelvin difference)

651 J/(kg °C) (joules per kilogram degree Celsius difference)
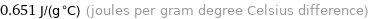
0.651 J/(g °C) (joules per gram degree Celsius difference)

0.156 BTU_IT/(lb °F) (IT British thermal units per pound degree Fahrenheit difference)
Comparisons for median specific heat at STP 651 J/(kg K)
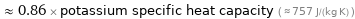
≈ 0.86 × potassium specific heat capacity ( ≈ 757 J/(kg K) )
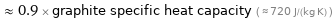
≈ 0.9 × graphite specific heat capacity ( ≈ 720 J/(kg K) )
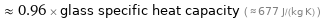
≈ 0.96 × glass specific heat capacity ( ≈ 677 J/(kg K) )
Thermodynamic properties
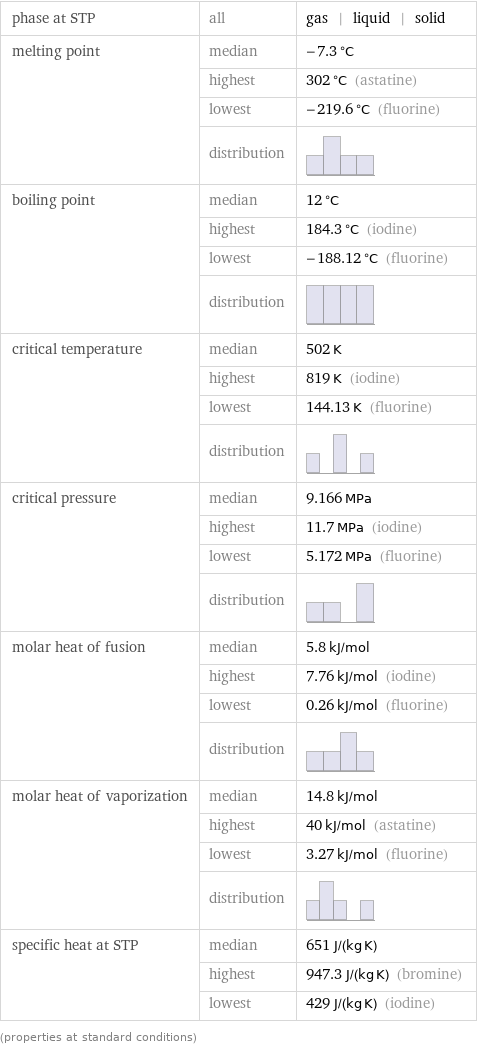
phase at STP | all | gas | liquid | solid melting point | median | -7.3 °C | highest | 302 °C (astatine) | lowest | -219.6 °C (fluorine) | distribution | boiling point | median | 12 °C | highest | 184.3 °C (iodine) | lowest | -188.12 °C (fluorine) | distribution | critical temperature | median | 502 K | highest | 819 K (iodine) | lowest | 144.13 K (fluorine) | distribution | critical pressure | median | 9.166 MPa | highest | 11.7 MPa (iodine) | lowest | 5.172 MPa (fluorine) | distribution | molar heat of fusion | median | 5.8 kJ/mol | highest | 7.76 kJ/mol (iodine) | lowest | 0.26 kJ/mol (fluorine) | distribution | molar heat of vaporization | median | 14.8 kJ/mol | highest | 40 kJ/mol (astatine) | lowest | 3.27 kJ/mol (fluorine) | distribution | specific heat at STP | median | 651 J/(kg K) | highest | 947.3 J/(kg K) (bromine) | lowest | 429 J/(kg K) (iodine) (properties at standard conditions)