Input interpretation
gallamine triethiodide
Chemical names and formulas
![formula | C_30H_60I_3N_3O_3 name | gallamine triethiodide IUPAC name | 2-[2, 6-bis(2-triethylammonioethoxy)phenoxy]ethyl-triethyl-azanium triiodide alternate names | benzcurine iodide | benzkurin mass fractions | C (carbon) 40.4% | H (hydrogen) 6.78% | I (iodine) 42.7% | N (nitrogen) 4.71% | O (oxygen) 5.38%](../image_source/26a3a9ad1184502d9b0a86467def6e8b.png)
formula | C_30H_60I_3N_3O_3 name | gallamine triethiodide IUPAC name | 2-[2, 6-bis(2-triethylammonioethoxy)phenoxy]ethyl-triethyl-azanium triiodide alternate names | benzcurine iodide | benzkurin mass fractions | C (carbon) 40.4% | H (hydrogen) 6.78% | I (iodine) 42.7% | N (nitrogen) 4.71% | O (oxygen) 5.38%
Structure diagram
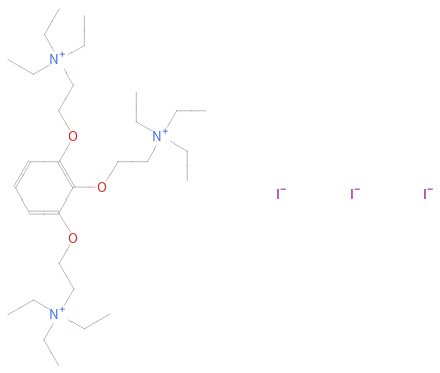
Structure diagram
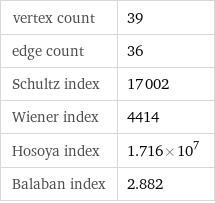
vertex count | 39 edge count | 36 Schultz index | 17002 Wiener index | 4414 Hosoya index | 1.716×10^7 Balaban index | 2.882
Basic properties
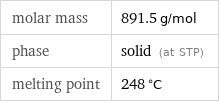
molar mass | 891.5 g/mol phase | solid (at STP) melting point | 248 °C
Units

Hydrophobicity and permeability properties

experimental LogP hydrophobicity | 3.5 predicted LogP hydrophobicity | 4.55 predicted LogS | -3.5
Basic drug properties

approval status | approved | small molecule drug categories | neuromuscular nondepolarizing agent | nicotinic antagonist | skeletal muscle relaxant

brand names | flacedil | flaxedil | fourneau 2559 | gallaflex | miowas G | parexyl | pirolakson | pyrolaxon | relaxan | remyolan | retensin | sincurarine | syncurarine | tricuran
Chemical identifiers
(CC)CCOC1=C(C(=CC=C1)OCC[N+](CC)(CC)CC)OCC[N+](CC)(CC)CC.[I-].[I-].[I-] InChI identifier | InChI=1/C30H60N3O3.3HI/c1-10-31(11-2, 12-3)22-25-34-28-20-19-21-29(35-26-23-32(13-4, 14-5)15-6)30(28)36-27-24-33(16-7, 17-8)18-9;;;/h19-21H, 10-18, 22-27H2, 1-9H3;3*1H/q+3;;;/p-3/fC30H60N3O3.3I/h;3*1h/qm;3*-1 InChI key | ICLWTJIMXVISSR-UHFFFAOYAV EU number | 200-605-1 RTECS number | BS1100000 NSC number | 102690](../image_source/dbc2c1b89c01468635d6e277ac26fee9.png)
CAS number | 65-29-2 Beilstein number | 3900826 PubChem CID number | 6172 PubChem SID number | 209540 SMILES identifier | CC[N+](CC)(CC)CCOC1=C(C(=CC=C1)OCC[N+](CC)(CC)CC)OCC[N+](CC)(CC)CC.[I-].[I-].[I-] InChI identifier | InChI=1/C30H60N3O3.3HI/c1-10-31(11-2, 12-3)22-25-34-28-20-19-21-29(35-26-23-32(13-4, 14-5)15-6)30(28)36-27-24-33(16-7, 17-8)18-9;;;/h19-21H, 10-18, 22-27H2, 1-9H3;3*1H/q+3;;;/p-3/fC30H60N3O3.3I/h;3*1h/qm;3*-1 InChI key | ICLWTJIMXVISSR-UHFFFAOYAV EU number | 200-605-1 RTECS number | BS1100000 NSC number | 102690
Toxicity properties

RTECS classes | drug