Input interpretation
aluminum chloride | nickel(II) chloride
Chemical names and formulas
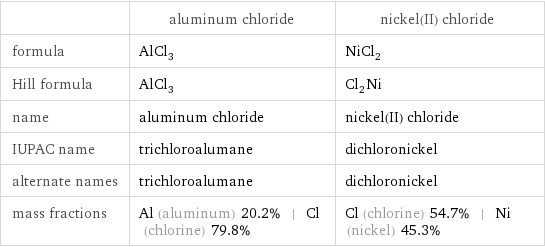
| aluminum chloride | nickel(II) chloride formula | AlCl_3 | NiCl_2 Hill formula | AlCl_3 | Cl_2Ni name | aluminum chloride | nickel(II) chloride IUPAC name | trichloroalumane | dichloronickel alternate names | trichloroalumane | dichloronickel mass fractions | Al (aluminum) 20.2% | Cl (chlorine) 79.8% | Cl (chlorine) 54.7% | Ni (nickel) 45.3%
Structure diagrams
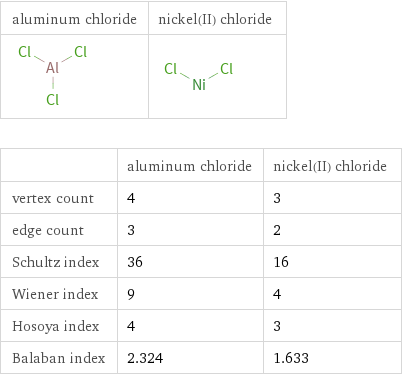
| aluminum chloride | nickel(II) chloride vertex count | 4 | 3 edge count | 3 | 2 Schultz index | 36 | 16 Wiener index | 9 | 4 Hosoya index | 4 | 3 Balaban index | 2.324 | 1.633
Basic properties
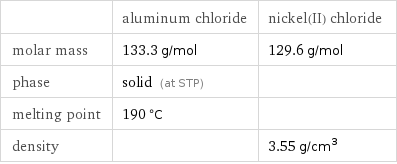
| aluminum chloride | nickel(II) chloride molar mass | 133.3 g/mol | 129.6 g/mol phase | solid (at STP) | melting point | 190 °C | density | | 3.55 g/cm^3
Units

Thermodynamic properties
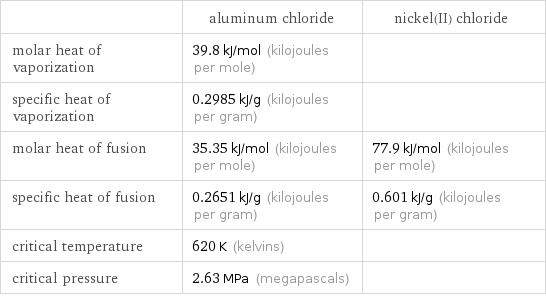
| aluminum chloride | nickel(II) chloride molar heat of vaporization | 39.8 kJ/mol (kilojoules per mole) | specific heat of vaporization | 0.2985 kJ/g (kilojoules per gram) | molar heat of fusion | 35.35 kJ/mol (kilojoules per mole) | 77.9 kJ/mol (kilojoules per mole) specific heat of fusion | 0.2651 kJ/g (kilojoules per gram) | 0.601 kJ/g (kilojoules per gram) critical temperature | 620 K (kelvins) | critical pressure | 2.63 MPa (megapascals) |
Solid properties

| aluminum chloride vapor pressure | 0.9998 mmHg
Units
