Input interpretation

silver(I) acetylide | molecular mass
Result

Find the molecular mass, m, for silver(I) acetylide: m = sum _iN_im_i Plan: • Write the chemical formula and gather atomic masses from the periodic table. • Determine values for N_i and m_i using these items. • Finally, compute the mass. Write the chemical formula: Ag_2C_2 Use the chemical formula, Ag_2C_2, to count the number of atoms, N_i, for each element: | N_i Ag (silver) | 2 C (carbon) | 2 Look up the atomic mass, m_i, in unified atomic mass units, u, for each element in the periodic table: | N_i | m_i/u Ag (silver) | 2 | 107.8682 C (carbon) | 2 | 12.011 Multiply N_i by m_i to compute the mass for each element. Then sum those values to compute the molecular mass, m: Answer: | | | N_i | m_i/u | mass/u Ag (silver) | 2 | 107.8682 | 2 × 107.8682 = 215.7364 C (carbon) | 2 | 12.011 | 2 × 12.011 = 24.022 m = 215.7364 u + 24.022 u = 239.758 u
Unit conversions
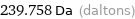
239.758 Da (daltons)
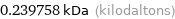
0.239758 kDa (kilodaltons)
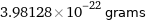
3.98128×10^-22 grams
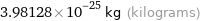
3.98128×10^-25 kg (kilograms)

239.769 chemical atomic mass units (unit officially deprecated)

239.835 physical atomic mass units (unit officially deprecated)
Comparisons as mass of molecule
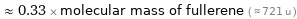
≈ 0.33 × molecular mass of fullerene ( ≈ 721 u )
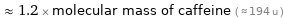
≈ 1.2 × molecular mass of caffeine ( ≈ 194 u )
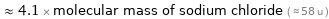
≈ 4.1 × molecular mass of sodium chloride ( ≈ 58 u )
Corresponding quantities
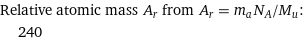
Relative atomic mass A_r from A_r = m_aN_A/M_u: | 240
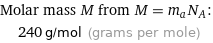
Molar mass M from M = m_aN_A: | 240 g/mol (grams per mole)
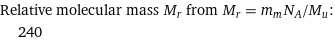
Relative molecular mass M_r from M_r = m_mN_A/M_u: | 240
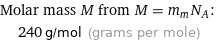
Molar mass M from M = m_mN_A: | 240 g/mol (grams per mole)