Input interpretation
calcium cation hydrogen carbonate anion | ionic radius
Results
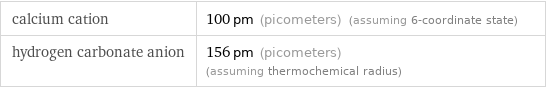
calcium cation | 100 pm (picometers) (assuming 6-coordinate state) hydrogen carbonate anion | 156 pm (picometers) (assuming thermochemical radius)
Ionic radii
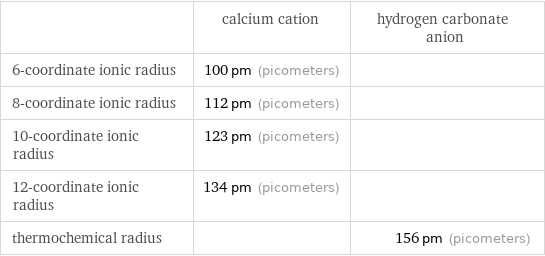
| calcium cation | hydrogen carbonate anion 6-coordinate ionic radius | 100 pm (picometers) | 8-coordinate ionic radius | 112 pm (picometers) | 10-coordinate ionic radius | 123 pm (picometers) | 12-coordinate ionic radius | 134 pm (picometers) | thermochemical radius | | 156 pm (picometers)