Input interpretation

elements | molar heat of vaporization
Summary

median | 272 kJ/mol highest | 800 kJ/mol (tungsten) lowest | 0.083 kJ/mol (helium) distribution | | (based on 94 values; 24 unavailable)
Entities with missing values

americium | curium | berkelium | californium | einsteinium | fermium | mendelevium | nobelium | lawrencium | rutherfordium | ... (total: 24)
Distribution plots
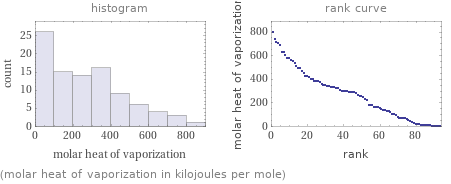
(molar heat of vaporization in kilojoules per mole)
Molar heat of vaporization rankings
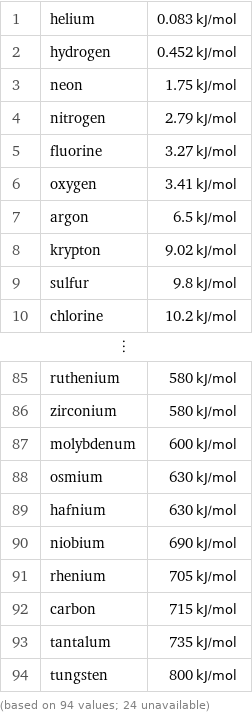
1 | helium | 0.083 kJ/mol 2 | hydrogen | 0.452 kJ/mol 3 | neon | 1.75 kJ/mol 4 | nitrogen | 2.79 kJ/mol 5 | fluorine | 3.27 kJ/mol 6 | oxygen | 3.41 kJ/mol 7 | argon | 6.5 kJ/mol 8 | krypton | 9.02 kJ/mol 9 | sulfur | 9.8 kJ/mol 10 | chlorine | 10.2 kJ/mol ⋮ | | 85 | ruthenium | 580 kJ/mol 86 | zirconium | 580 kJ/mol 87 | molybdenum | 600 kJ/mol 88 | osmium | 630 kJ/mol 89 | hafnium | 630 kJ/mol 90 | niobium | 690 kJ/mol 91 | rhenium | 705 kJ/mol 92 | carbon | 715 kJ/mol 93 | tantalum | 735 kJ/mol 94 | tungsten | 800 kJ/mol (based on 94 values; 24 unavailable)
Unit conversions for median molar heat of vaporization 272 kJ/mol
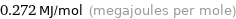
0.272 MJ/mol (megajoules per mole)
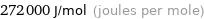
272000 J/mol (joules per mole)

65.1 kcal/mol (thermochemical kilocalories per mole)
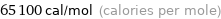
65100 cal/mol (calories per mole)
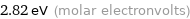
2.82 eV (molar electronvolts)