Input interpretation

alkaline earth metals | number of protons
Summary
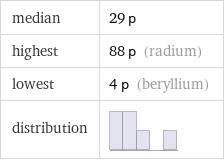
median | 29 p highest | 88 p (radium) lowest | 4 p (beryllium) distribution |
Units

Ranked values

| | visual | ratios | 6 | beryllium | | 0.04545 | 1 5 | magnesium | | 0.1364 | 3 4 | calcium | | 0.2273 | 5 3 | strontium | | 0.4318 | 9.5 2 | barium | | 0.6364 | 14 1 | radium | | 1 | 22
Number of protons rankings
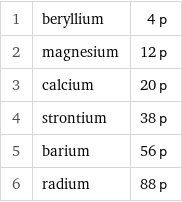
1 | beryllium | 4 p 2 | magnesium | 12 p 3 | calcium | 20 p 4 | strontium | 38 p 5 | barium | 56 p 6 | radium | 88 p
Corresponding quantity
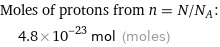
Moles of protons from n = N/N_A: | 4.8×10^-23 mol (moles)