Input interpretation
1 cm^3 of pearl ash
Basic properties for 1 cm^3

mass | 2.43 grams 0.00243 kg (kilograms) molar amount | 17.6 mmol (millimoles) 0.0176 mol (moles) volume | 1 mL (milliliter) 1000 µL (microliters) 0.001 L (liters) 1×10^-6 m^3 (cubic meters) 0.203 tsp (teaspoons) 0.00106 quarts equivalents | 0.035 eq (equivalents) of potassium cation 0.018 eq (equivalents) of carbonate anion (at STP)
Corresponding quantities

sphere radius | 6.204 mm (millimeters) side of a cube | 10 mm (millimeters)
Thermodynamic properties for 1 cm^3
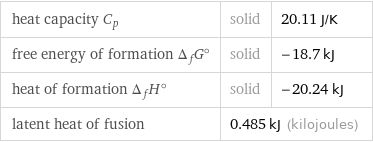
heat capacity C_p | solid | 20.11 J/K free energy of formation Δ_fG° | solid | -18.7 kJ heat of formation Δ_fH° | solid | -20.24 kJ latent heat of fusion | 0.485 kJ (kilojoules) |
Units

Phase change energies for 1 cm^3 from 25 °C

energy required to heat to melting point | 17.4 kJ (kilojoules) energy required to convert to liquid | 0.485 kJ (kilojoules) energy required to heat to melting point and convert to liquid | 17.9 kJ (kilojoules)
Mass composition for 1 cm^3
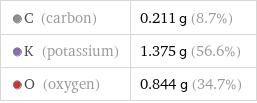
C (carbon) | 0.211 g (8.7%) K (potassium) | 1.375 g (56.6%) O (oxygen) | 0.844 g (34.7%)
Mass composition for 1 cm^3
Structure diagram
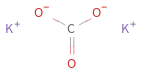
Structure diagram
vertex count | 6 edge count | 3 Schultz index | 36 Wiener index | 9 Hosoya index | 4 Balaban index | 2.324
Chemical names and formulas
formula | K_2CO_3 Hill formula | CK_2O_3 name | pearl ash IUPAC name | dipotassium carbonate
Substance properties
molar mass | 138.2 g/mol phase | solid (at STP) melting point | 891 °C density | 2.43 g/cm^3 solubility in water | soluble
Units
