Input interpretation

noble metals | specific heat at STP
Summary
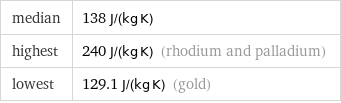
median | 138 J/(kg K) highest | 240 J/(kg K) (rhodium and palladium) lowest | 129.1 J/(kg K) (gold)
Units

Distribution plots
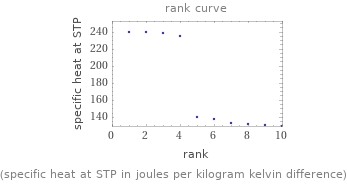
(specific heat at STP in joules per kilogram kelvin difference)
Specific heat at STP rankings
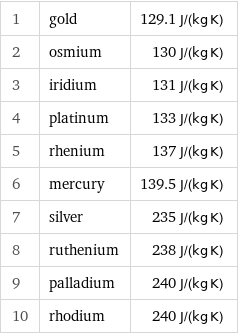
1 | gold | 129.1 J/(kg K) 2 | osmium | 130 J/(kg K) 3 | iridium | 131 J/(kg K) 4 | platinum | 133 J/(kg K) 5 | rhenium | 137 J/(kg K) 6 | mercury | 139.5 J/(kg K) 7 | silver | 235 J/(kg K) 8 | ruthenium | 238 J/(kg K) 9 | palladium | 240 J/(kg K) 10 | rhodium | 240 J/(kg K)
Unit conversions for median specific heat at STP 138 J/(kg K)
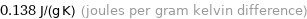
0.138 J/(g K) (joules per gram kelvin difference)
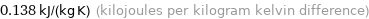
0.138 kJ/(kg K) (kilojoules per kilogram kelvin difference)
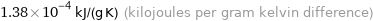
1.38×10^-4 kJ/(g K) (kilojoules per gram kelvin difference)

138 J/(kg °C) (joules per kilogram degree Celsius difference)
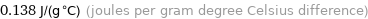
0.138 J/(g °C) (joules per gram degree Celsius difference)

0.033 BTU_IT/(lb °F) (IT British thermal units per pound degree Fahrenheit difference)
Comparisons for median specific heat at STP 138 J/(kg K)
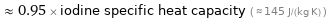
≈ 0.95 × iodine specific heat capacity ( ≈ 145 J/(kg K) )
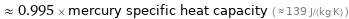
≈ 0.995 × mercury specific heat capacity ( ≈ 139 J/(kg K) )
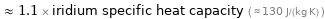
≈ 1.1 × iridium specific heat capacity ( ≈ 130 J/(kg K) )
Thermodynamic properties

phase at STP | all | liquid | solid melting point | median | 1866 °C | highest | 3186 °C (rhenium) | lowest | -38.83 °C (mercury) | distribution | boiling point | median | 3760 °C | highest | 5596 °C (rhenium) | lowest | 356.73 °C (mercury) | distribution | molar heat of fusion | median | 21 kJ/mol | highest | 33 kJ/mol (rhenium) | lowest | 2.29 kJ/mol (mercury) | distribution | molar heat of vaporization | median | 493 kJ/mol | highest | 705 kJ/mol (rhenium) | lowest | 59.2 kJ/mol (mercury) | distribution | specific heat at STP | median | 138 J/(kg K) | highest | 240 J/(kg K) (rhodium and palladium) | lowest | 129.1 J/(kg K) (gold) (properties at standard conditions)