Input interpretation
phosphite anion | nitrate anion | nitrite anion
Structure diagrams
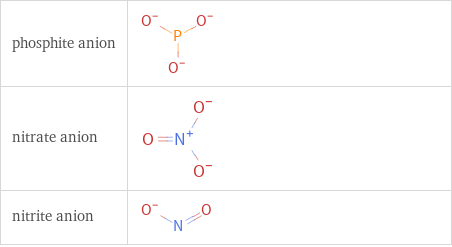
Structure diagrams
General properties
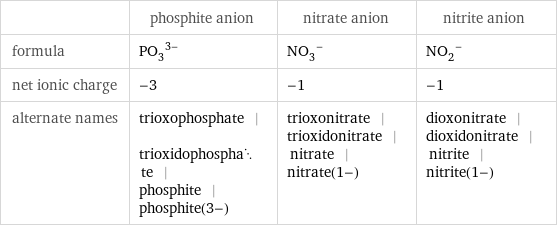
| phosphite anion | nitrate anion | nitrite anion formula | (PO_3)^(3-) | (NO_3)^- | (NO_2)^- net ionic charge | -3 | -1 | -1 alternate names | trioxophosphate | trioxidophosphate | phosphite | phosphite(3-) | trioxonitrate | trioxidonitrate | nitrate | nitrate(1-) | dioxonitrate | dioxidonitrate | nitrite | nitrite(1-)
Ionic radii

| nitrate anion | nitrite anion thermochemical radius | 179 pm | 192 pm
Units

Other properties
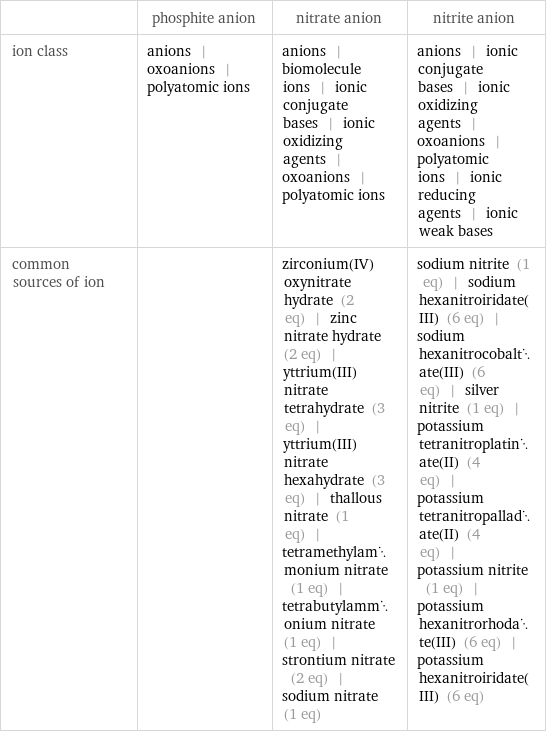
| phosphite anion | nitrate anion | nitrite anion ion class | anions | oxoanions | polyatomic ions | anions | biomolecule ions | ionic conjugate bases | ionic oxidizing agents | oxoanions | polyatomic ions | anions | ionic conjugate bases | ionic oxidizing agents | oxoanions | polyatomic ions | ionic reducing agents | ionic weak bases common sources of ion | | zirconium(IV) oxynitrate hydrate (2 eq) | zinc nitrate hydrate (2 eq) | yttrium(III)nitrate tetrahydrate (3 eq) | yttrium(III) nitrate hexahydrate (3 eq) | thallous nitrate (1 eq) | tetramethylammonium nitrate (1 eq) | tetrabutylammonium nitrate (1 eq) | strontium nitrate (2 eq) | sodium nitrate (1 eq) | sodium nitrite (1 eq) | sodium hexanitroiridate(III) (6 eq) | sodium hexanitrocobaltate(III) (6 eq) | silver nitrite (1 eq) | potassium tetranitroplatinate(II) (4 eq) | potassium tetranitropalladate(II) (4 eq) | potassium nitrite (1 eq) | potassium hexanitrorhodate(III) (6 eq) | potassium hexanitroiridate(III) (6 eq)