Input interpretation
sertraline | L-serine
Chemical names and formulas
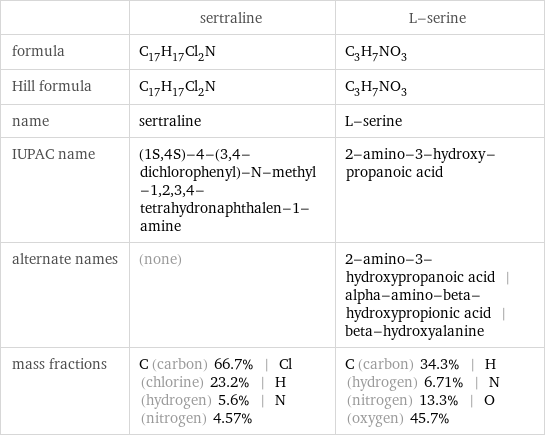
| sertraline | L-serine formula | C_17H_17Cl_2N | C_3H_7NO_3 Hill formula | C_17H_17Cl_2N | C_3H_7NO_3 name | sertraline | L-serine IUPAC name | (1S, 4S)-4-(3, 4-dichlorophenyl)-N-methyl-1, 2, 3, 4-tetrahydronaphthalen-1-amine | 2-amino-3-hydroxy-propanoic acid alternate names | (none) | 2-amino-3-hydroxypropanoic acid | alpha-amino-beta-hydroxypropionic acid | beta-hydroxyalanine mass fractions | C (carbon) 66.7% | Cl (chlorine) 23.2% | H (hydrogen) 5.6% | N (nitrogen) 4.57% | C (carbon) 34.3% | H (hydrogen) 6.71% | N (nitrogen) 13.3% | O (oxygen) 45.7%
Structure diagrams
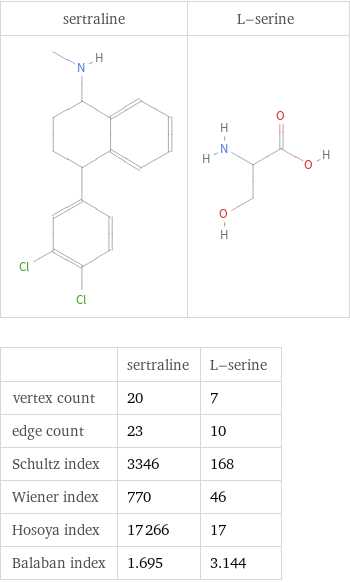
| sertraline | L-serine vertex count | 20 | 7 edge count | 23 | 10 Schultz index | 3346 | 168 Wiener index | 770 | 46 Hosoya index | 17266 | 17 Balaban index | 1.695 | 3.144
3D structure
3D structure
Basic properties
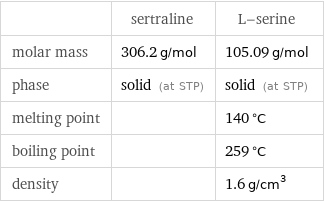
| sertraline | L-serine molar mass | 306.2 g/mol | 105.09 g/mol phase | solid (at STP) | solid (at STP) melting point | | 140 °C boiling point | | 259 °C density | | 1.6 g/cm^3
Units

Hydrophobicity and permeability properties
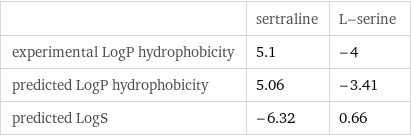
| sertraline | L-serine experimental LogP hydrophobicity | 5.1 | -4 predicted LogP hydrophobicity | 5.06 | -3.41 predicted LogS | -6.32 | 0.66
Amino acid properties
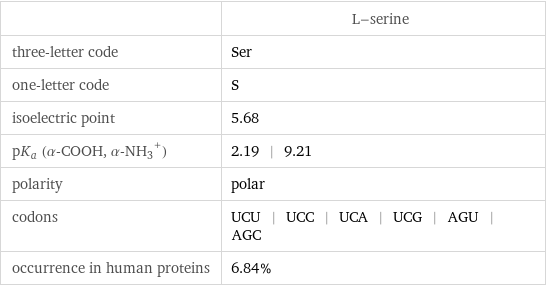
| L-serine three-letter code | Ser one-letter code | S isoelectric point | 5.68 pK_a (α-COOH, (α-NH_3)^+) | 2.19 | 9.21 polarity | polar codons | UCU | UCC | UCA | UCG | AGU | AGC occurrence in human proteins | 6.84%