Input interpretation

p-menth-4(8)-ene | aromatic atom count
Result

0 atoms
Aromatic atoms in place
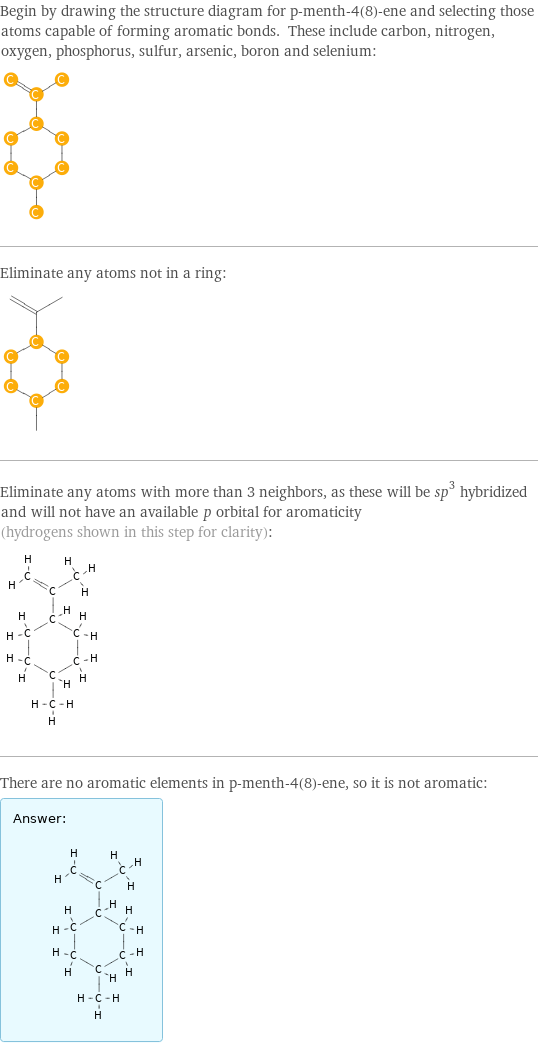 "Italic"]\) orbital for aromaticity (hydrogens shown in this step for clarity): There are no aromatic elements in p-menth-4(8)-ene, so it is not aromatic: Answer: | | " />
"Italic"]\) orbital for aromaticity (hydrogens shown in this step for clarity): There are no aromatic elements in p-menth-4(8)-ene, so it is not aromatic: Answer: | | " />
Begin by drawing the structure diagram for p-menth-4(8)-ene and selecting those atoms capable of forming aromatic bonds. These include carbon, nitrogen, oxygen, phosphorus, sulfur, arsenic, boron and selenium: Eliminate any atoms not in a ring: Eliminate any atoms with more than 3 neighbors, as these will be sp^3 hybridized and will not have an available \!\(\*StyleBox["p", FontSlant->"Italic"]\) orbital for aromaticity (hydrogens shown in this step for clarity): There are no aromatic elements in p-menth-4(8)-ene, so it is not aromatic: Answer: | |
Quantitative molecular descriptors
longest chain length | 7 atoms longest straight chain length | 3 atoms longest aliphatic chain length | 3 atoms aromatic atom count | 0 atoms H-bond acceptor count | 0 atoms H-bond donor count | 0 atoms