Input interpretation
beryllium sulfate
Chemical names and formulas

formula | BeSO_4 Hill formula | BeO_4S name | beryllium sulfate IUPAC name | beryllium(+2) cation sulfate alternate names | beryllium sulfate(1:1) | beryllium sulphate | sulfuric acid, beryllium salt (1:1) mass fractions | Be (beryllium) 8.58% | O (oxygen) 60.9% | S (sulfur) 30.5%
Structure diagram
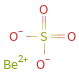
Structure diagram
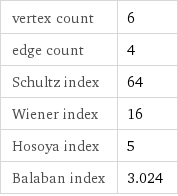
vertex count | 6 edge count | 4 Schultz index | 64 Wiener index | 16 Hosoya index | 5 Balaban index | 3.024
Basic properties
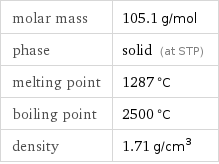
molar mass | 105.1 g/mol phase | solid (at STP) melting point | 1287 °C boiling point | 2500 °C density | 1.71 g/cm^3
Units

Solid properties (at STP)
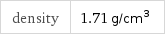
density | 1.71 g/cm^3
Units

Thermodynamic properties
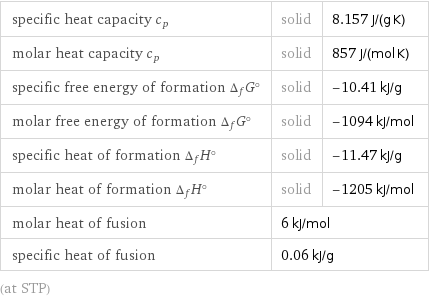
specific heat capacity c_p | solid | 8.157 J/(g K) molar heat capacity c_p | solid | 857 J/(mol K) specific free energy of formation Δ_fG° | solid | -10.41 kJ/g molar free energy of formation Δ_fG° | solid | -1094 kJ/mol specific heat of formation Δ_fH° | solid | -11.47 kJ/g molar heat of formation Δ_fH° | solid | -1205 kJ/mol molar heat of fusion | 6 kJ/mol | specific heat of fusion | 0.06 kJ/g | (at STP)
Chemical identifiers
![CAS number | 13510-49-1 PubChem CID number | 26077 SMILES identifier | [Be+2].[O-]S(=O)(=O)[O-] InChI identifier | InChI=1/Be.H2O4S/c;1-5(2, 3)4/h;(H2, 1, 2, 3, 4)/q+2;/p-2/fBe.O4S/qm;-2 EU number | 236-842-2 Gmelin number | 8296 RTECS number | DS4800000 NSC number | 84266](../image_source/946dbc1f0df5e8efc8245812fecf3340.png)
CAS number | 13510-49-1 PubChem CID number | 26077 SMILES identifier | [Be+2].[O-]S(=O)(=O)[O-] InChI identifier | InChI=1/Be.H2O4S/c;1-5(2, 3)4/h;(H2, 1, 2, 3, 4)/q+2;/p-2/fBe.O4S/qm;-2 EU number | 236-842-2 Gmelin number | 8296 RTECS number | DS4800000 NSC number | 84266
Toxicity properties

odor | odorless lethal dosage | 82 mg/kg (oral dose for rats)

probable lethal dose for man | 30 mL (milliliters) RTECS classes | tumorigen | mutagen
Ion equivalents

Be^(2+) (beryllium cation) | 1 (SO_4)^(2-) (sulfate anion) | 1