Input interpretation

mineral acids | molar mass
Summary
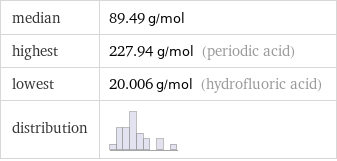
median | 89.49 g/mol highest | 227.94 g/mol (periodic acid) lowest | 20.006 g/mol (hydrofluoric acid) distribution |
Units

Distribution plots
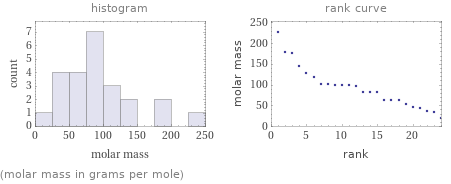
(molar mass in grams per mole)
Molar mass rankings
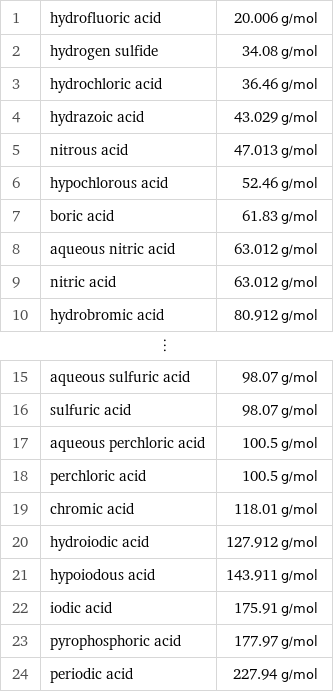
1 | hydrofluoric acid | 20.006 g/mol 2 | hydrogen sulfide | 34.08 g/mol 3 | hydrochloric acid | 36.46 g/mol 4 | hydrazoic acid | 43.029 g/mol 5 | nitrous acid | 47.013 g/mol 6 | hypochlorous acid | 52.46 g/mol 7 | boric acid | 61.83 g/mol 8 | aqueous nitric acid | 63.012 g/mol 9 | nitric acid | 63.012 g/mol 10 | hydrobromic acid | 80.912 g/mol ⋮ | | 15 | aqueous sulfuric acid | 98.07 g/mol 16 | sulfuric acid | 98.07 g/mol 17 | aqueous perchloric acid | 100.5 g/mol 18 | perchloric acid | 100.5 g/mol 19 | chromic acid | 118.01 g/mol 20 | hydroiodic acid | 127.912 g/mol 21 | hypoiodous acid | 143.911 g/mol 22 | iodic acid | 175.91 g/mol 23 | pyrophosphoric acid | 177.97 g/mol 24 | periodic acid | 227.94 g/mol
Unit conversion for median molar mass 89.49 g/mol
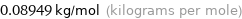
0.08949 kg/mol (kilograms per mole)
Comparisons for median molar mass 89.49 g/mol
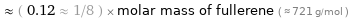
≈ ( 0.12 ≈ 1/8 ) × molar mass of fullerene ( ≈ 721 g/mol )
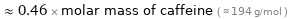
≈ 0.46 × molar mass of caffeine ( ≈ 194 g/mol )
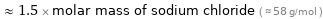
≈ 1.5 × molar mass of sodium chloride ( ≈ 58 g/mol )
Corresponding quantities
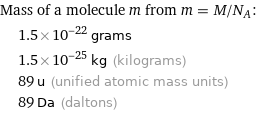
Mass of a molecule m from m = M/N_A: | 1.5×10^-22 grams | 1.5×10^-25 kg (kilograms) | 89 u (unified atomic mass units) | 89 Da (daltons)
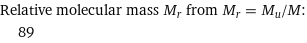
Relative molecular mass M_r from M_r = M_u/M: | 89