Input interpretation

aluminum (chemical element)
Periodic table location
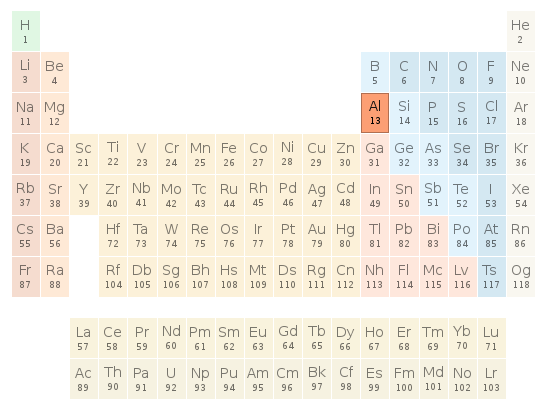
Periodic table location
Image

Image
Basic elemental properties
![atomic symbol | Al atomic number | 13 short electronic configuration | [Ne]3s^23p^1 Aufbau diagram | 3p 3s block | p group | 13 period | 3 atomic mass | 26.9815385 u (unified atomic mass units)](../image_source/623636490f83621cdd3c08acd0fc5180.png)
atomic symbol | Al atomic number | 13 short electronic configuration | [Ne]3s^23p^1 Aufbau diagram | 3p 3s block | p group | 13 period | 3 atomic mass | 26.9815385 u (unified atomic mass units)
Thermodynamic properties

phase at STP | solid melting point | 660.32 °C (rank: 65th) boiling point | 2519 °C (rank: 48th) molar heat of fusion | 10.7 kJ/mol (rank: 46th) molar heat of vaporization | 293 kJ/mol (rank: 42nd) specific heat at STP | 904 J/(kg K) (rank: 12th) (properties at standard conditions)
Material properties
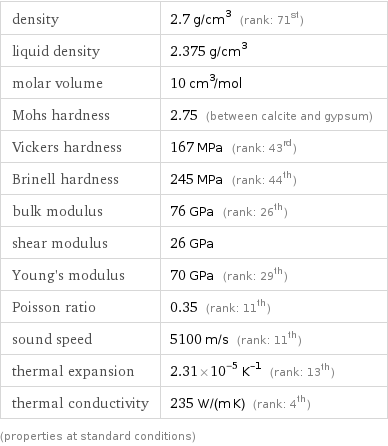
density | 2.7 g/cm^3 (rank: 71st) liquid density | 2.375 g/cm^3 molar volume | 10 cm^3/mol Mohs hardness | 2.75 (between calcite and gypsum) Vickers hardness | 167 MPa (rank: 43rd) Brinell hardness | 245 MPa (rank: 44th) bulk modulus | 76 GPa (rank: 26th) shear modulus | 26 GPa Young's modulus | 70 GPa (rank: 29th) Poisson ratio | 0.35 (rank: 11th) sound speed | 5100 m/s (rank: 11th) thermal expansion | 2.31×10^-5 K^(-1) (rank: 13th) thermal conductivity | 235 W/(m K) (rank: 4th) (properties at standard conditions)
Electromagnetic properties
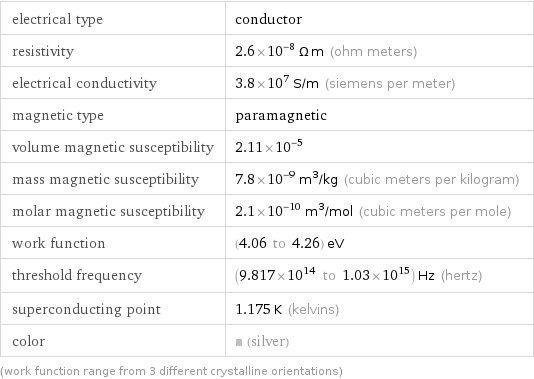
electrical type | conductor resistivity | 2.6×10^-8 Ω m (ohm meters) electrical conductivity | 3.8×10^7 S/m (siemens per meter) magnetic type | paramagnetic volume magnetic susceptibility | 2.11×10^-5 mass magnetic susceptibility | 7.8×10^-9 m^3/kg (cubic meters per kilogram) molar magnetic susceptibility | 2.1×10^-10 m^3/mol (cubic meters per mole) work function | (4.06 to 4.26) eV threshold frequency | (9.817×10^14 to 1.03×10^15) Hz (hertz) superconducting point | 1.175 K (kelvins) color | (silver) (work function range from 3 different crystalline orientations)
Reactivity
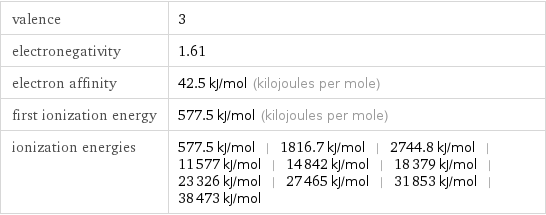
valence | 3 electronegativity | 1.61 electron affinity | 42.5 kJ/mol (kilojoules per mole) first ionization energy | 577.5 kJ/mol (kilojoules per mole) ionization energies | 577.5 kJ/mol | 1816.7 kJ/mol | 2744.8 kJ/mol | 11577 kJ/mol | 14842 kJ/mol | 18379 kJ/mol | 23326 kJ/mol | 27465 kJ/mol | 31853 kJ/mol | 38473 kJ/mol
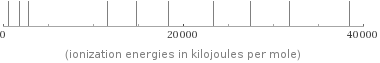
Reactivity
Atomic properties

term symbol | ^2P_(1/2) atomic radius | 125 pm covalent radius | 121 pm (electronic ground state properties)
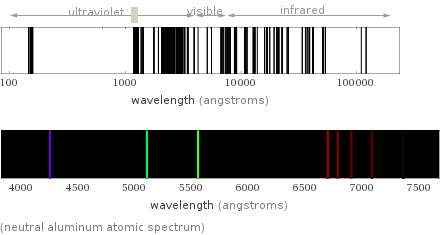
(neutral aluminum atomic spectrum)
Abundances
universe abundance | 0.005 mass% (rank: 14th) solar abundance | 0.006 mass% (rank: 14th) meteorite abundance | 0.91 mass% (rank: 10th) crust abundance | 8.1 mass% (rank: 3rd) ocean abundance | 5×10^-7 mass% (rank: 23rd) human abundance | 9×10^-5 mass% (rank: 21st) universe molar abundance | 2×10^-4 mol% (rank: 13th) solar molar abundance | 3×10^-4 mol% (rank: 11th) meteorite molar abundance | 0.67 mol% (rank: 8th) ocean molar abundance | 1.1×10^-7 mol% (rank: 33rd) crust molar abundance | 6.3 mol% (rank: 3rd) human molar abundance | 2.1×10^-5 mol% (rank: 20th)
Nuclear properties
half-life | (stable) stable isotopes | Al-27 (100%) nuclear spin | Al-21: 5/2^+ | Al-22: 3^+ | Al-23: 5/2^+ | Al-24: 4^+ | Al-25: 5/2^+ | Al-26: 5^+ | Al-27: 5/2^+ | Al-28: 3^+ | Al-29: 5/2^+ | Al-30: 3^+ | Al-32: 1^+ | Al-33: 5/2^+ | Al-34: 4^- | Al-35: 5/2^+ | Al-37: 3/2^+ | Al-39: 3/2^+ | Al-41: 3/2^+ unstable isotopes | Al-26 (0.717 Myr) | Al-29 (6.57 min) | Al-28 (134.48 s) | Al-25 (7.183 s) | Al-30 (3.6 s) | Al-24 (2.053 s) | Al-31 (644 ms) | Al-23 (470 ms) | Al-36 (90 ms) | Al-22 (59 ms) | Al-34 (42 ms) | Al-33 (41.7 ms) | Al-35 (38.6 ms) | Al-32 (33 ms) | Al-37 (10.7 ms) | Al-38 (7.6 ms) | Al-39 (7.6 µs) | Al-41 (260 ns) | Al-40 (260 ns) | Al-42 (170 ns) | Al-21 (35 ns) neutron cross-section | 0.233 b neutron mass absorption | 0.03 m^2/kg
Identifiers

CAS number | 7429-90-5 PubChem CID number | 5359268