Input interpretation
Lewis bases
Members
hydrogen | ammonia | water | carbon monoxide | phosphine | hydrogen sulfide | trimethylamine | dimethyl sulfide | benzene | N, N, N', N'-tetramethylethylenediamine (total: 10)
Structure diagrams
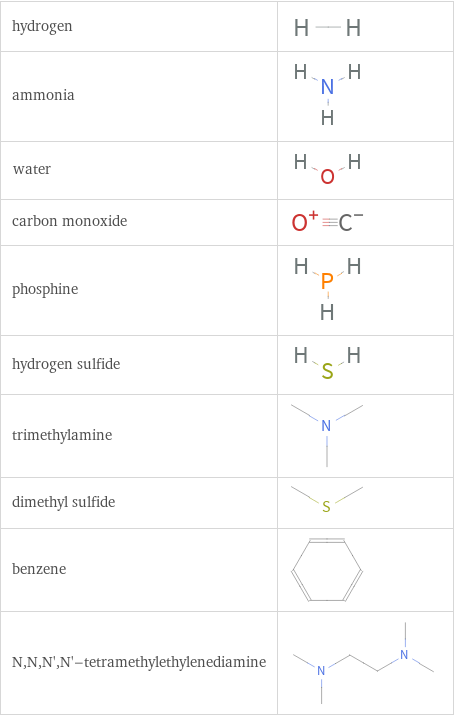
Structure diagrams
Basic properties

molar mass | median | 34.04 g/mol | highest | 116.21 g/mol (N, N, N', N'-tetramethylethylenediamine) | lowest | 2.016 g/mol (hydrogen) | distribution | melting point | median | -91.5 °C | highest | 5.5 °C (benzene) | lowest | -259.2 °C (hydrogen) | distribution | boiling point | median | -14.92 °C | highest | 121 °C (N, N, N', N'-tetramethylethylenediamine) | lowest | -252.8 °C (hydrogen) | distribution | density | median | 0.3157 g/cm^3 | highest | 1 g/cm^3 (water) | lowest | 8.99×10^-5 g/cm^3 (hydrogen) | distribution |
Units
