Input interpretation
germanium dioxide
Chemical names and formulas

formula | GeO_2 name | germanium dioxide alternate names | diketogermane | dioxogermane | germania | germanic acid | germanic oxide (crystalline) | germanium oxide mass fractions | Ge (germanium) 69.4% | O (oxygen) 30.6%
Structure diagram
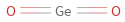
Structure diagram
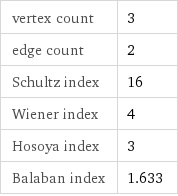
vertex count | 3 edge count | 2 Schultz index | 16 Wiener index | 4 Hosoya index | 3 Balaban index | 1.633
Basic properties
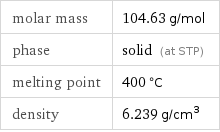
molar mass | 104.63 g/mol phase | solid (at STP) melting point | 400 °C density | 6.239 g/cm^3
Units

Solid properties (at STP)
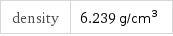
density | 6.239 g/cm^3
Units

Thermodynamic properties
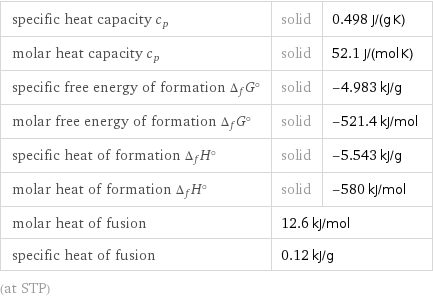
specific heat capacity c_p | solid | 0.498 J/(g K) molar heat capacity c_p | solid | 52.1 J/(mol K) specific free energy of formation Δ_fG° | solid | -4.983 kJ/g molar free energy of formation Δ_fG° | solid | -521.4 kJ/mol specific heat of formation Δ_fH° | solid | -5.543 kJ/g molar heat of formation Δ_fH° | solid | -580 kJ/mol molar heat of fusion | 12.6 kJ/mol | specific heat of fusion | 0.12 kJ/g | (at STP)
Chemical identifiers
=O RTECS number | LY5240000 MDL number | MFCD00011030](../image_source/3a89826744821621e76ce5546ae2f31a.png)
CAS number | 1310-53-8 SMILES identifier | [Ge](=O)=O RTECS number | LY5240000 MDL number | MFCD00011030
Toxicity properties

RTECS classes | reproductive effector | human data