Input interpretation
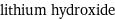
lithium hydroxide
Chemical names and formulas

formula | LiOH Hill formula | HLiO name | lithium hydroxide alternate names | lithium hydrate | lithium hydroxide (1:1) | lithium hydroxide anhydrous | lithium hydroxide, solution mass fractions | H (hydrogen) 4.21% | Li (lithium) 29% | O (oxygen) 66.8%
Structure diagram

Structure diagram
vertex count | 2 edge count | 1 Schultz index | 0 Wiener index | 0 Hosoya index | 1 Balaban index | 0
Basic properties
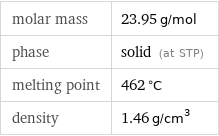
molar mass | 23.95 g/mol phase | solid (at STP) melting point | 462 °C density | 1.46 g/cm^3
Units

Solid properties (at STP)

density | 1.46 g/cm^3 vapor pressure | 2 mmHg (at 520 °C)
Units

Thermodynamic properties
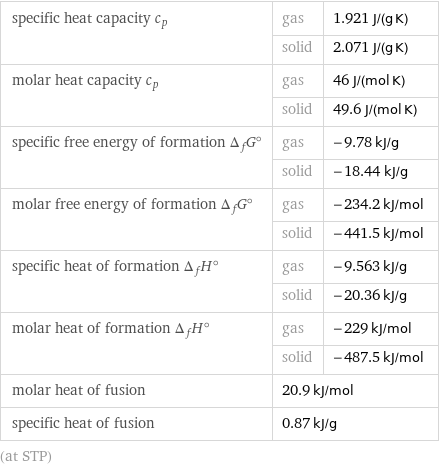
specific heat capacity c_p | gas | 1.921 J/(g K) | solid | 2.071 J/(g K) molar heat capacity c_p | gas | 46 J/(mol K) | solid | 49.6 J/(mol K) specific free energy of formation Δ_fG° | gas | -9.78 kJ/g | solid | -18.44 kJ/g molar free energy of formation Δ_fG° | gas | -234.2 kJ/mol | solid | -441.5 kJ/mol specific heat of formation Δ_fH° | gas | -9.563 kJ/g | solid | -20.36 kJ/g molar heat of formation Δ_fH° | gas | -229 kJ/mol | solid | -487.5 kJ/mol molar heat of fusion | 20.9 kJ/mol | specific heat of fusion | 0.87 kJ/g | (at STP)
Chemical identifiers
![CAS number | 1310-65-2 PubChem CID number | 3939 SMILES identifier | [Li+].[OH-] InChI identifier | InChI=1/Li.H2O/h;1H2/q+1;/p-1/fLi.HO/h;1h/qm;-1 EU number | 215-183-4 Gmelin number | 19032 RTECS number | OJ6307070](../image_source/ca1a9f13150bfa21fa49ff84904dbf15.png)
CAS number | 1310-65-2 PubChem CID number | 3939 SMILES identifier | [Li+].[OH-] InChI identifier | InChI=1/Li.H2O/h;1H2/q+1;/p-1/fLi.HO/h;1h/qm;-1 EU number | 215-183-4 Gmelin number | 19032 RTECS number | OJ6307070
NFPA label

NFPA label

NFPA health rating | 3 NFPA fire rating | 0 NFPA reactivity rating | 0
Toxicity properties

odor | odorless

RTECS classes | other
Ion equivalents

Li^+ (lithium cation) | 1 (OH)^- (hydroxide anion) | 1