Input interpretation

complex ions | molar mass
Summary
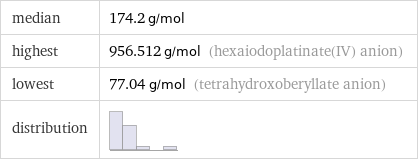
median | 174.2 g/mol highest | 956.512 g/mol (hexaiodoplatinate(IV) anion) lowest | 77.04 g/mol (tetrahydroxoberyllate anion) distribution |
Units

Distribution plots
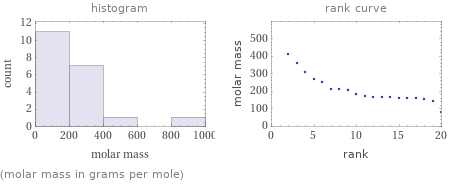
(molar mass in grams per mole)
Molar mass rankings
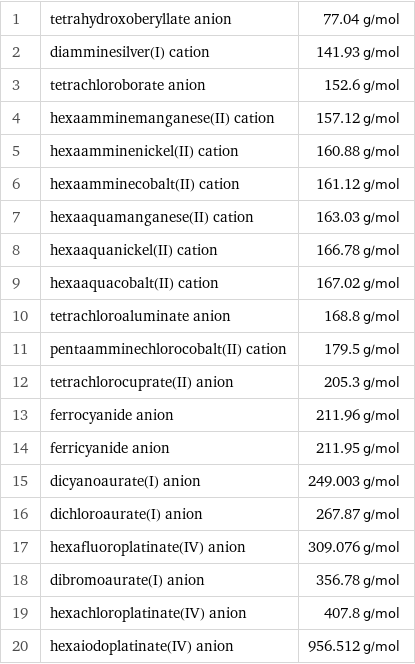
1 | tetrahydroxoberyllate anion | 77.04 g/mol 2 | diamminesilver(I) cation | 141.93 g/mol 3 | tetrachloroborate anion | 152.6 g/mol 4 | hexaamminemanganese(II) cation | 157.12 g/mol 5 | hexaamminenickel(II) cation | 160.88 g/mol 6 | hexaamminecobalt(II) cation | 161.12 g/mol 7 | hexaaquamanganese(II) cation | 163.03 g/mol 8 | hexaaquanickel(II) cation | 166.78 g/mol 9 | hexaaquacobalt(II) cation | 167.02 g/mol 10 | tetrachloroaluminate anion | 168.8 g/mol 11 | pentaamminechlorocobalt(II) cation | 179.5 g/mol 12 | tetrachlorocuprate(II) anion | 205.3 g/mol 13 | ferrocyanide anion | 211.96 g/mol 14 | ferricyanide anion | 211.95 g/mol 15 | dicyanoaurate(I) anion | 249.003 g/mol 16 | dichloroaurate(I) anion | 267.87 g/mol 17 | hexafluoroplatinate(IV) anion | 309.076 g/mol 18 | dibromoaurate(I) anion | 356.78 g/mol 19 | hexachloroplatinate(IV) anion | 407.8 g/mol 20 | hexaiodoplatinate(IV) anion | 956.512 g/mol
Unit conversion for median molar mass 174.2 g/mol
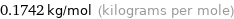
0.1742 kg/mol (kilograms per mole)
Comparisons for median molar mass 174.2 g/mol
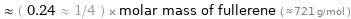
≈ ( 0.24 ≈ 1/4 ) × molar mass of fullerene ( ≈ 721 g/mol )
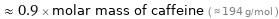
≈ 0.9 × molar mass of caffeine ( ≈ 194 g/mol )
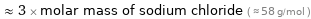
≈ 3 × molar mass of sodium chloride ( ≈ 58 g/mol )
Corresponding quantities
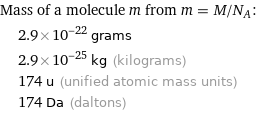
Mass of a molecule m from m = M/N_A: | 2.9×10^-22 grams | 2.9×10^-25 kg (kilograms) | 174 u (unified atomic mass units) | 174 Da (daltons)
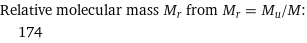
Relative molecular mass M_r from M_r = M_u/M: | 174