Input interpretation
period 1 elements
Members
hydrogen | helium
Periodic table location
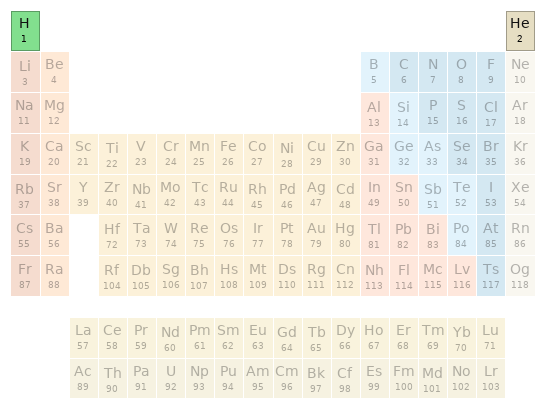
Periodic table location
Images

Images
Basic elemental properties
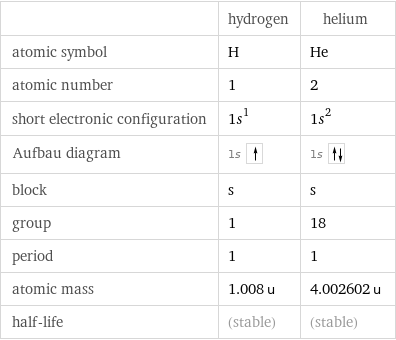
| hydrogen | helium atomic symbol | H | He atomic number | 1 | 2 short electronic configuration | 1s^1 | 1s^2 Aufbau diagram | 1s | 1s block | s | s group | 1 | 18 period | 1 | 1 atomic mass | 1.008 u | 4.002602 u half-life | (stable) | (stable)
Thermodynamic properties
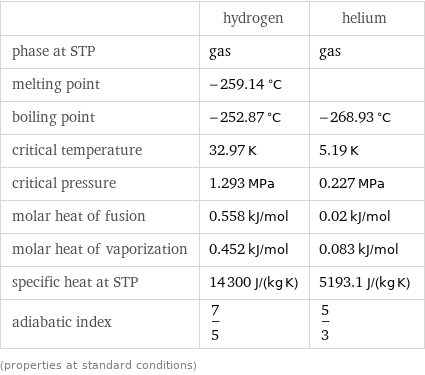
| hydrogen | helium phase at STP | gas | gas melting point | -259.14 °C | boiling point | -252.87 °C | -268.93 °C critical temperature | 32.97 K | 5.19 K critical pressure | 1.293 MPa | 0.227 MPa molar heat of fusion | 0.558 kJ/mol | 0.02 kJ/mol molar heat of vaporization | 0.452 kJ/mol | 0.083 kJ/mol specific heat at STP | 14300 J/(kg K) | 5193.1 J/(kg K) adiabatic index | 7/5 | 5/3 (properties at standard conditions)
Material properties
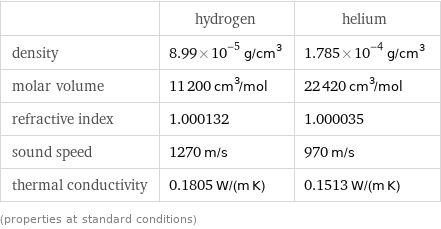
| hydrogen | helium density | 8.99×10^-5 g/cm^3 | 1.785×10^-4 g/cm^3 molar volume | 11200 cm^3/mol | 22420 cm^3/mol refractive index | 1.000132 | 1.000035 sound speed | 1270 m/s | 970 m/s thermal conductivity | 0.1805 W/(m K) | 0.1513 W/(m K) (properties at standard conditions)
Electromagnetic properties
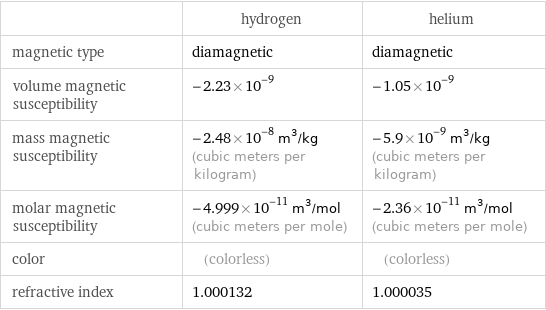
| hydrogen | helium magnetic type | diamagnetic | diamagnetic volume magnetic susceptibility | -2.23×10^-9 | -1.05×10^-9 mass magnetic susceptibility | -2.48×10^-8 m^3/kg (cubic meters per kilogram) | -5.9×10^-9 m^3/kg (cubic meters per kilogram) molar magnetic susceptibility | -4.999×10^-11 m^3/mol (cubic meters per mole) | -2.36×10^-11 m^3/mol (cubic meters per mole) color | (colorless) | (colorless) refractive index | 1.000132 | 1.000035
Reactivity
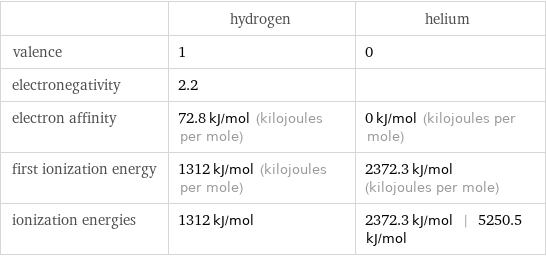
| hydrogen | helium valence | 1 | 0 electronegativity | 2.2 | electron affinity | 72.8 kJ/mol (kilojoules per mole) | 0 kJ/mol (kilojoules per mole) first ionization energy | 1312 kJ/mol (kilojoules per mole) | 2372.3 kJ/mol (kilojoules per mole) ionization energies | 1312 kJ/mol | 2372.3 kJ/mol | 5250.5 kJ/mol
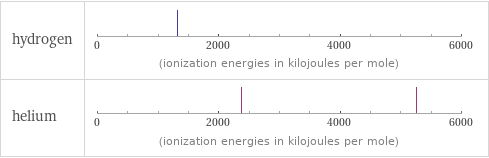
Reactivity
Atomic properties

| hydrogen | helium term symbol | ^2S_(1/2) | ^1S_0 atomic radius | 25 pm | 31 pm covalent radius | 31 pm | 28 pm van der Waals radius | 120 pm | 140 pm (electronic ground state properties)
Abundances
| hydrogen | helium universe abundance | 75 mass% | 23 mass% solar abundance | 75 mass% | 23 mass% meteorite abundance | 2.4 mass% | crust abundance | 0.15 mass% | 5.5×10^-7 mass% ocean abundance | 11 mass% | 7.2×10^-10 mass% human abundance | 10 mass% | universe molar abundance | 93 mol% | 7.2 mol% solar molar abundance | 93 mol% | 7.4 mol% meteorite molar abundance | 17 mol% | ocean molar abundance | 66 mol% | 1.1×10^-9 mol% crust molar abundance | 3.1 mol% | 3×10^-6 mol% human molar abundance | 62 mol% |
Nuclear properties
| hydrogen | helium half-life | (stable) | (stable) stable isotopes | H-1 (99.985%) | H-2 (1.5×10^-4) | He-4 (99.999863%) | He-3 (1.37×10^-6) nuclear spin | H-1: 1/2^+ | H-2: 1^+ | H-3: 1/2^+ | H-4: 2^- | H-5: 1/2^+ | H-6: 2^- | H-7: 1/2^+ | He-3: 1/2^+ | He-4: 0^+ | He-5: 3/2^- | He-6: 0^+ | He-7: 3/2^- | He-8: 0^+ | He-9: 1/2^+ | He-10: 0^+ unstable isotopes | H-3 (12.32 yr) | H-6 (0.29 zs) | H-4 (0.139 zs) | H-5 (0.08 zs) | H-7 (0.023 zs) | He-6 (806.7 ms) | He-8 (119 ms) | He-9 (7 zs) | He-7 (3.04 zs) | He-10 (1.52 zs) | He-5 (0.76 zs) neutron cross-section | 0.332 b | 0.007 b neutron mass absorption | 0.011 m^2/kg | 1×10^-5 m^2/kg
Identifiers
| hydrogen | helium CAS number | 1333-74-0 | 7440-59-7 PubChem CID number | 783 | 23987