Input interpretation

top 10 chemicals | by vapor pressure
Result

1 | acetylene | 9.29×10^6 Pa | 2 | carbon dioxide | 5.725×10^6 Pa | 3 | ozone | 5.5009×10^6 Pa | 4 | phosphorus trifluoride | 5.44×10^6 Pa | 5 | 1-chloro-2-butene | 4.9441×10^6 Pa | 6 | methane | 4.52×10^6 Pa | 7 | fluoromethane-13 C | 4.479×10^6 Pa | 8 | fluoromethane-d 3 | 4.479×10^6 Pa | 9 | fluoroform | 4.377×10^6 Pa | 10 | hydrogen chloride | 4.225×10^6 Pa |
Structure diagrams
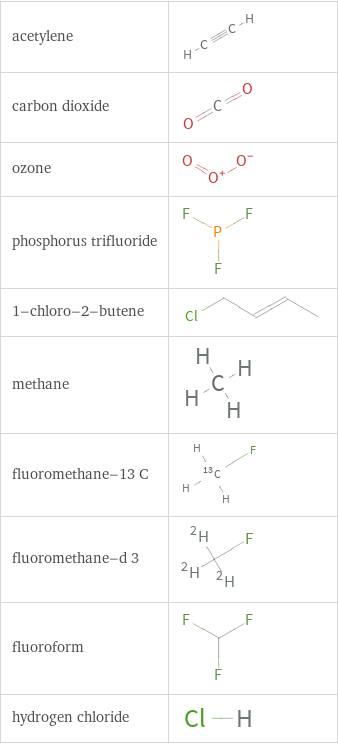
Structure diagrams
Basic properties

molar mass | median | 40.53 g/mol | highest | 90.55 g/mol (1-chloro-2-butene) | lowest | 16.04 g/mol (methane) | distribution | melting point | median | -141.8 °C | highest | -56.56 °C (carbon dioxide) | lowest | -192.2 °C (ozone) | distribution | boiling point | median | -81.75 °C | highest | 83 °C (1-chloro-2-butene) | lowest | -161.48 °C (methane) | distribution | density | median | 0.001962 g/cm^3 | highest | 0.9491 g/cm^3 (1-chloro-2-butene) | lowest | 6.67151×10^-4 g/cm^3 (methane) | distribution |
Units
