Input interpretation

oxides | molar mass
Summary
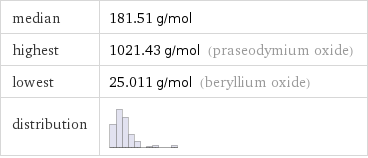
median | 181.51 g/mol highest | 1021.43 g/mol (praseodymium oxide) lowest | 25.011 g/mol (beryllium oxide) distribution |
Units

Distribution plots
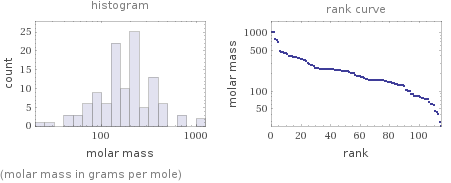
(molar mass in grams per mole)
Molar mass rankings
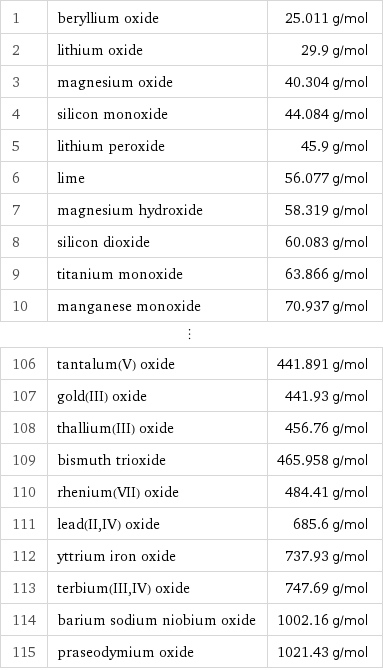
1 | beryllium oxide | 25.011 g/mol 2 | lithium oxide | 29.9 g/mol 3 | magnesium oxide | 40.304 g/mol 4 | silicon monoxide | 44.084 g/mol 5 | lithium peroxide | 45.9 g/mol 6 | lime | 56.077 g/mol 7 | magnesium hydroxide | 58.319 g/mol 8 | silicon dioxide | 60.083 g/mol 9 | titanium monoxide | 63.866 g/mol 10 | manganese monoxide | 70.937 g/mol ⋮ | | 106 | tantalum(V) oxide | 441.891 g/mol 107 | gold(III) oxide | 441.93 g/mol 108 | thallium(III) oxide | 456.76 g/mol 109 | bismuth trioxide | 465.958 g/mol 110 | rhenium(VII) oxide | 484.41 g/mol 111 | lead(II, IV) oxide | 685.6 g/mol 112 | yttrium iron oxide | 737.93 g/mol 113 | terbium(III, IV) oxide | 747.69 g/mol 114 | barium sodium niobium oxide | 1002.16 g/mol 115 | praseodymium oxide | 1021.43 g/mol
Unit conversion for median molar mass 181.51 g/mol
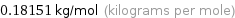
0.18151 kg/mol (kilograms per mole)
Comparisons for median molar mass 181.51 g/mol
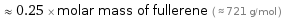
≈ 0.25 × molar mass of fullerene ( ≈ 721 g/mol )
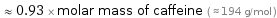
≈ 0.93 × molar mass of caffeine ( ≈ 194 g/mol )
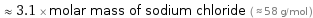
≈ 3.1 × molar mass of sodium chloride ( ≈ 58 g/mol )
Corresponding quantities

Mass of a molecule m from m = M/N_A: | 3×10^-22 grams | 3×10^-25 kg (kilograms) | 182 u (unified atomic mass units) | 182 Da (daltons)
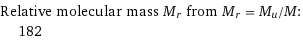
Relative molecular mass M_r from M_r = M_u/M: | 182