Input interpretation

period 2 elements | electron affinity
Summary
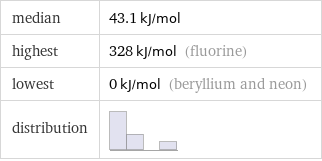
median | 43.1 kJ/mol highest | 328 kJ/mol (fluorine) lowest | 0 kJ/mol (beryllium and neon) distribution |
Units

Plot
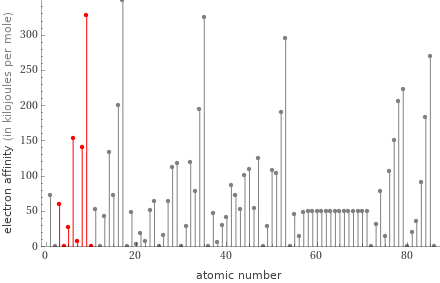
Plot
Periodic table location
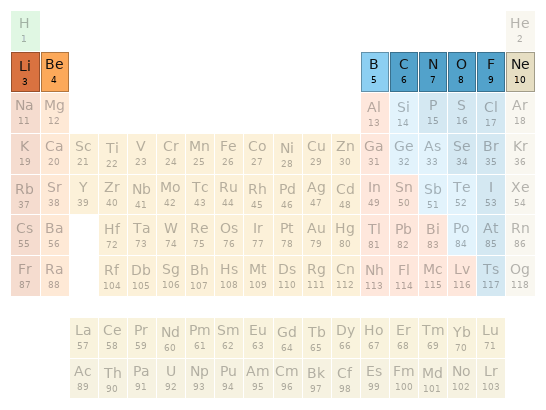
Periodic table location
Electron affinity rankings
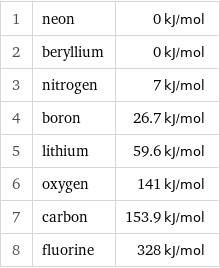
1 | neon | 0 kJ/mol 2 | beryllium | 0 kJ/mol 3 | nitrogen | 7 kJ/mol 4 | boron | 26.7 kJ/mol 5 | lithium | 59.6 kJ/mol 6 | oxygen | 141 kJ/mol 7 | carbon | 153.9 kJ/mol 8 | fluorine | 328 kJ/mol
Unit conversions for median electron affinity 43.1 kJ/mol
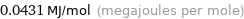
0.0431 MJ/mol (megajoules per mole)
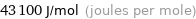
43100 J/mol (joules per mole)

10.3 kcal/mol (thermochemical kilocalories per mole)
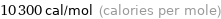
10300 cal/mol (calories per mole)

0.447 eV (molar electronvolts)