Input interpretation
red phosphorus | copper
Chemical names and formulas

| red phosphorus | copper formula | P | Cu Hill formula | P | Cu name | red phosphorus | copper IUPAC name | phosphorus | copper alternate names | amorphous phosphorus | phosphorus red | red amorphous phosphorus | copper metal mass fractions | P (phosphorus) 100% | Cu (copper) 100%
Structure diagrams

Structure diagrams
Basic properties

| red phosphorus | copper molar mass | 30.973761998 g/mol | 63.546 g/mol phase | solid (at STP) | solid (at STP) melting point | 579.2 °C | 1083 °C boiling point | | 2567 °C density | 2.16 g/cm^3 | 8.96 g/cm^3 solubility in water | insoluble | insoluble
Units

Thermodynamic properties
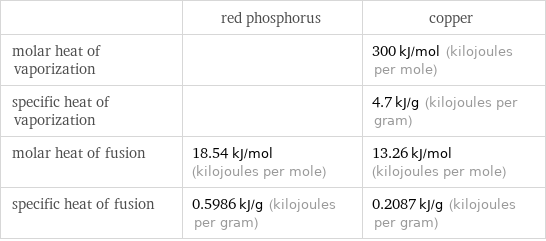
| red phosphorus | copper molar heat of vaporization | | 300 kJ/mol (kilojoules per mole) specific heat of vaporization | | 4.7 kJ/g (kilojoules per gram) molar heat of fusion | 18.54 kJ/mol (kilojoules per mole) | 13.26 kJ/mol (kilojoules per mole) specific heat of fusion | 0.5986 kJ/g (kilojoules per gram) | 0.2087 kJ/g (kilojoules per gram)
Solid properties (at STP)

| red phosphorus | copper density | 2.16 g/cm^3 | 8.96 g/cm^3 vapor pressure | 0.02999 mmHg | 1 mmHg
Units

Chemical identifiers
![| red phosphorus | copper CAS number | 7723-14-0 | 7440-50-8 PubChem CID number | 5462309 | 23978 PubChem SID number | 50725881 | 24852148 SMILES identifier | [P] | [Cu]](../image_source/5bbe2b347122fab1e1173cabd7857b0b.png)
| red phosphorus | copper CAS number | 7723-14-0 | 7440-50-8 PubChem CID number | 5462309 | 23978 PubChem SID number | 50725881 | 24852148 SMILES identifier | [P] | [Cu]